- 翰林提供學術活動、國際課程、科研項目一站式留學背景提升服務!
- 021-63526630
CIE A Level Chemistry復習筆記1.7.11 Strength of Acids & Bases
Strong & Weak Acids & Bases
- Strong and weak acids can be distinguished from each other by their:
- pH value?(using a pH meter or universal indicator)
- Electrical conductivity
- Reactivity
pH
- An acid?dissociates?into H+?in solution according to:
HA → H+?+ A-
- The?stronger?the acid, the?greater?the?concentration of H+?and therefore the?lower?the?pH
pH value of a strong acid & base table

- The most?accurate?way to determine the pH is by reading it off a?pH meter
- The pH meter is connected to the?pH electrode?which shows the pH value of the solution
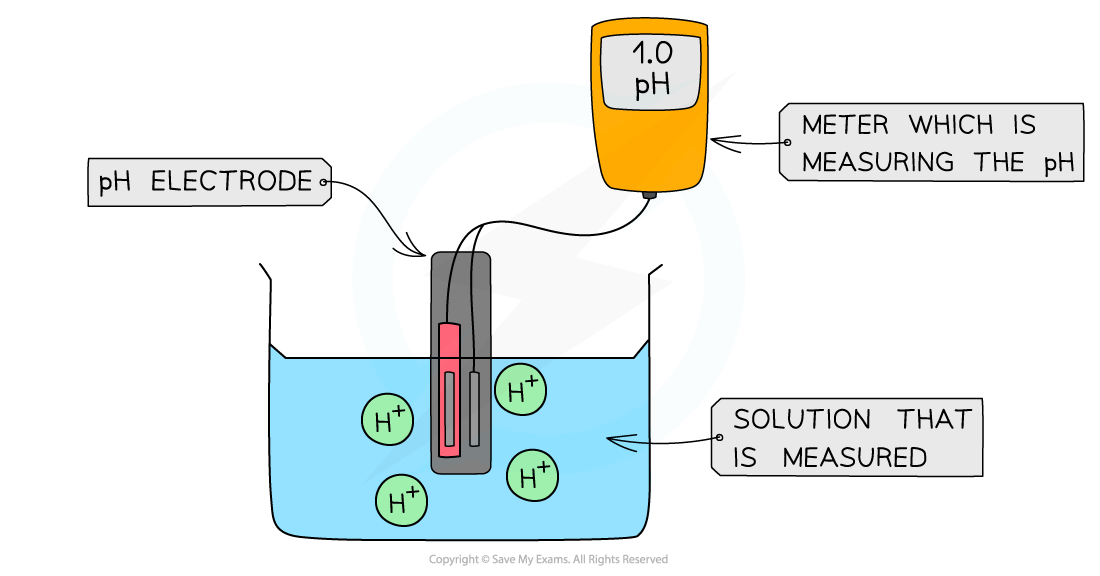
The diagram shows a digital pH meter that measures the pH of a solution using a pH electrode
- A less accurate method is to measure the pH using universal indicator paper
- The universal indicator paper is dipped into a solution of acid upon which the paper changes colour
- The colour is then compared to those on a chart which shows the colours corresponding to different pH values
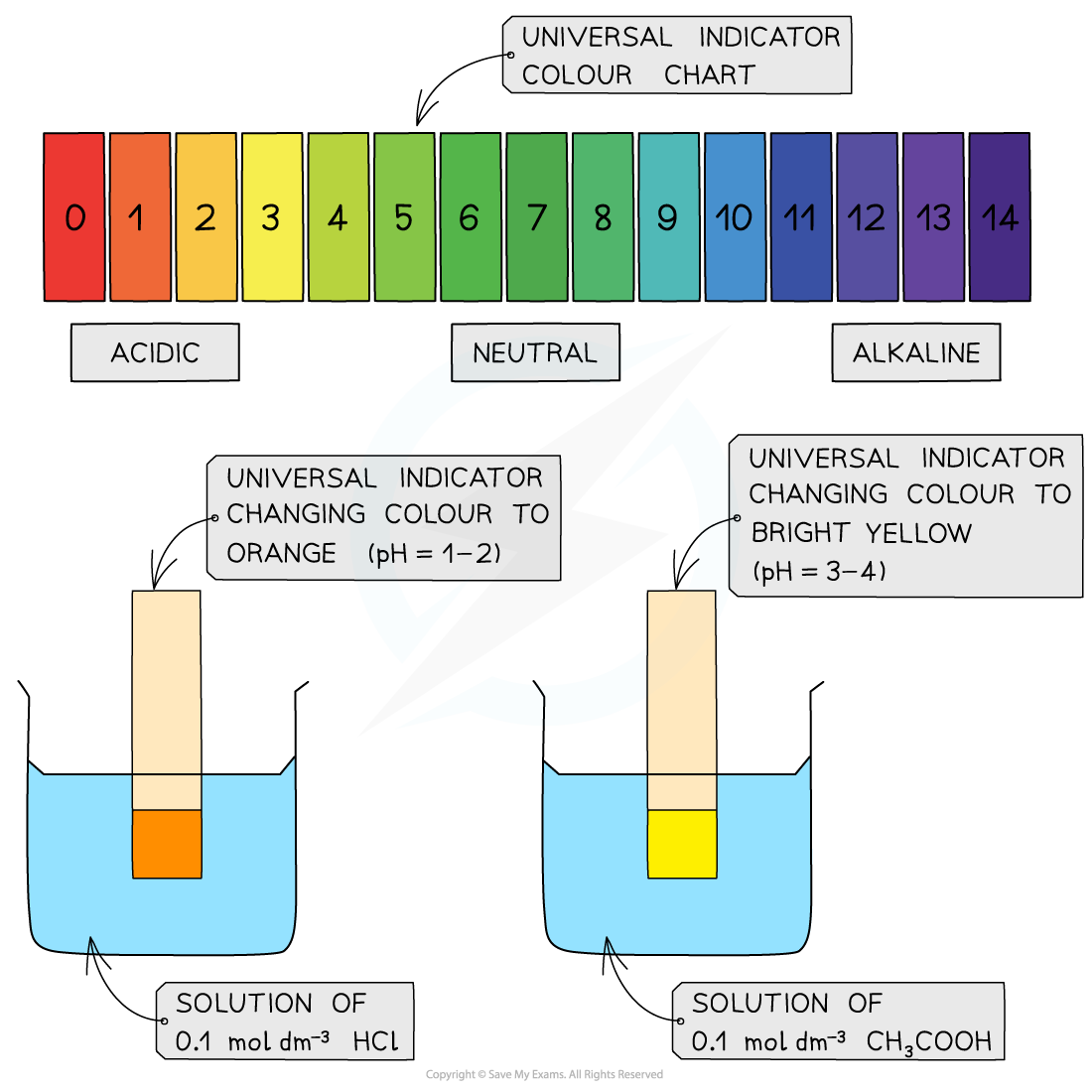
The diagram shows the change in colour of the universal indicator paper when dipped in a strong (HCl) and weak (CH3COOH) acid. The colour chart is used to read off the corresponding pH values which are between 1-2 for HCl and 3-4 for CH3COOH
Electrical conductivity
- Since a?stronger acid?has a?higher concentration of H+?it?conducts electricity?better
- Stronger acids therefore have a greater?electrical conductivity
- The electrical conductivity can be determined by using a?conductivity meter
- Like the pH meter, the conductivity meter is connected to an electrode
- The conductivity of the solution can be read off the meter
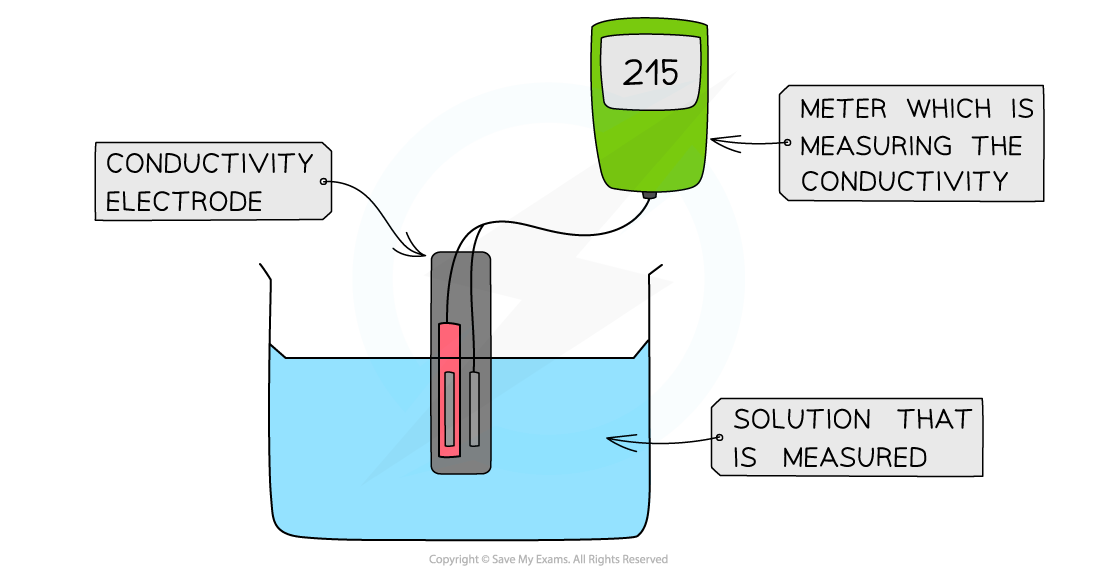 The diagram shows a digital conductivity meter that measures the electrical conductivity of a solution using an electrode
The diagram shows a digital conductivity meter that measures the electrical conductivity of a solution using an electrode
Reactivity
- Strong and weak acids of the same concentrations react differently with reactive metals
- This is because the concentration of H+?is greater in strong acids compared to weak acids
- The greater H+?concentration means that more H2?gas is produced
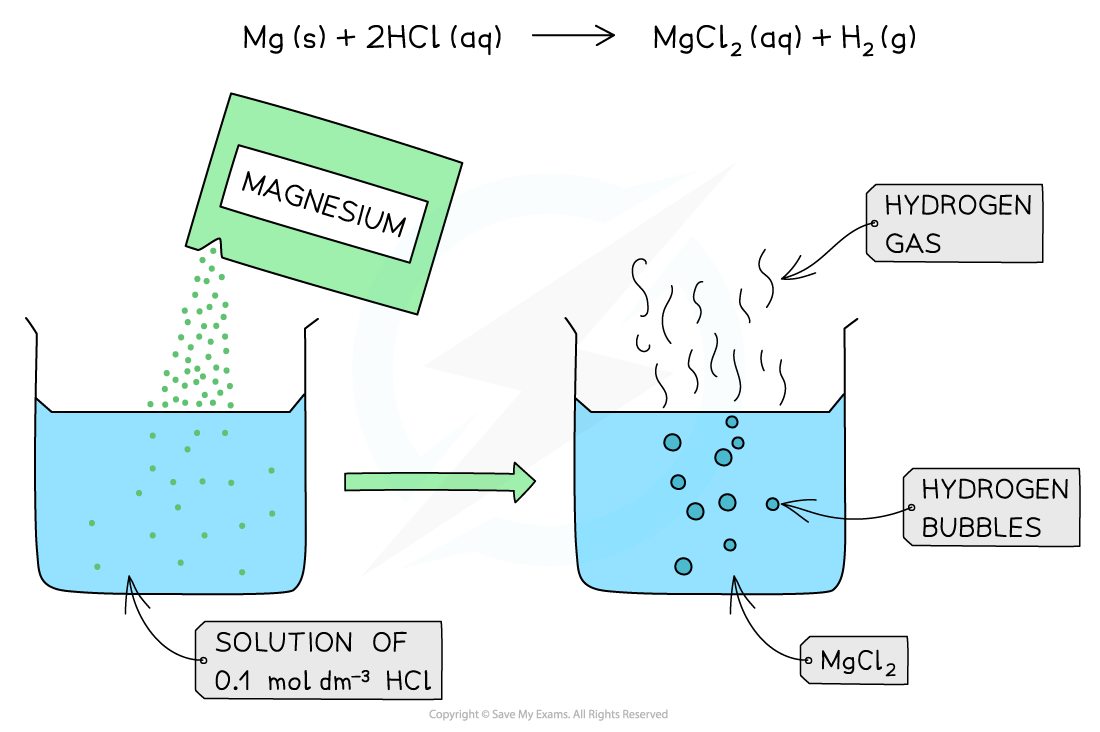 The diagram shows the reaction of 0.1 mol dm-3?of a strong acid (HCl) with Mg. The reaction produces a lot of bubbles and hydrogen gas due to the high concentration of H+?present in solution
The diagram shows the reaction of 0.1 mol dm-3?of a strong acid (HCl) with Mg. The reaction produces a lot of bubbles and hydrogen gas due to the high concentration of H+?present in solution
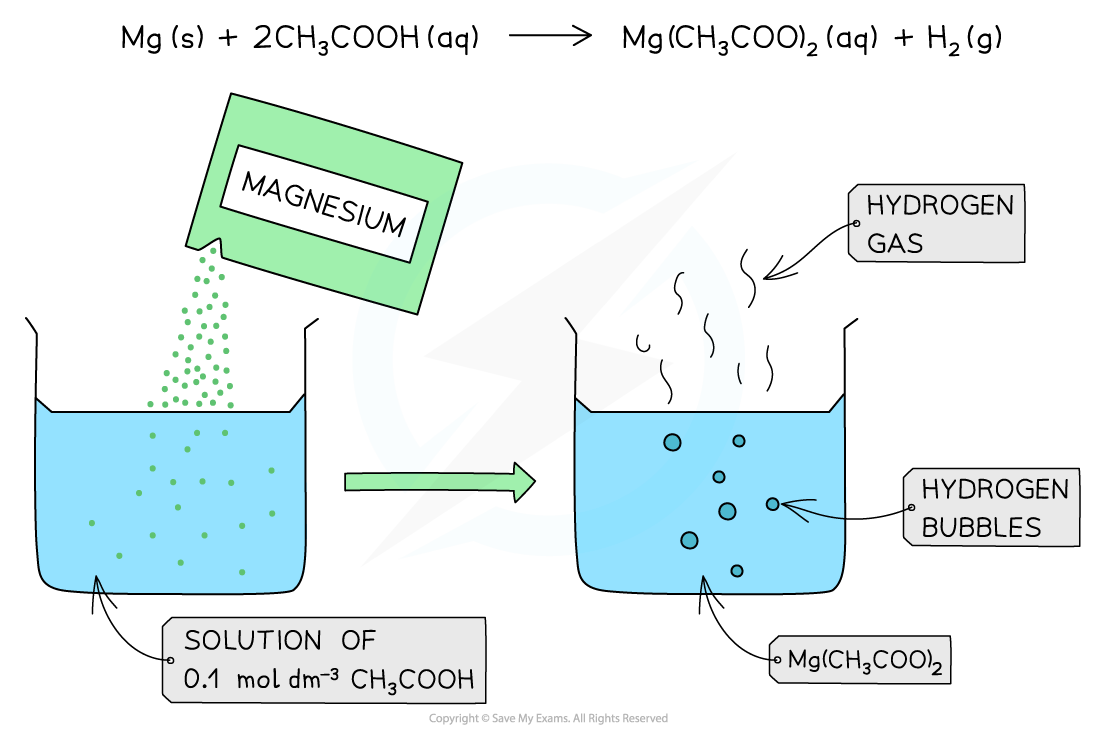
The diagram shows the reaction of 0.1 mol dm-3?of a weak acid (CH3COOH) with Mg. The reaction produces less bubbles and hydrogen gas due to the lower concentration of H+?present in solution
Exam Tip
The above-mentioned properties of strong and weak acids depend on their ability to dissociate and form H+?ions.Stronger acids dissociate more, producing a greater concentration of H+?ions and therefore showing lower pH values, greater electrical conductivity and more vigorous reactions with reactive metals.
Neutralisation Reactions
- A neutralisation reaction is one in which an acid (pH <7) and a base/alkali (pH >7) react together to form water (pH = 7) and a salt

- The proton of the acid reacts with the hydroxide of the base to form water

- The spectator ions which are not involved in the formation of water, form the salt
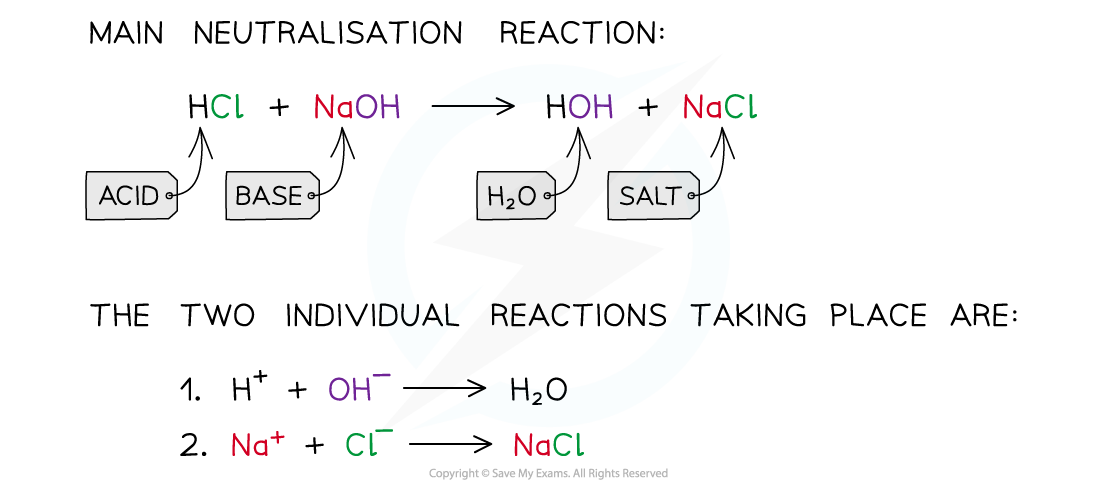
The diagram shows a neutralisation reaction of HCl and NaOH and the two individual reactions that take place to form the water and salt
- The name of the salt produced can be predicted from the acid that has reacted
Acid reacted & salt table

Exam Tip
Note that the reaction of an acid and metal carbonate also forms carbon dioxide:
acid + metal carbonate → salt + water + carbon dioxide
轉載自savemyexams
以上就是關于【CIE A Level Chemistry復習筆記1.7.11 Strength of Acids & Bases】的解答,如需了解學校/賽事/課程動態,可至翰林教育官網獲取更多信息。
往期文章閱讀推薦:
全網破防!ALevel CIE數學M1疑似錯題?經濟P2難度飆升?5月6日大考考情分析必看!
A-Level CIE就大規模泄題發布最嚴處罰!哪些考生必須重考?你的成績怎么辦?

最新發布
? 2026. All Rights Reserved. 滬ICP備2023009024號-1









