- 翰林提供學術活動、國際課程、科研項目一站式留學背景提升服務!
- 021-63526630
CIE A Level Chemistry復習筆記1.7.10 pH Scale
The pH Scale
- The pH scale is a numerical scale that shows how?acidic?or?alkaline?a solution is
- The values on the pH scale go from 1-14 (extremely acidic substances have values of below 1)
- All acids have pH values of?below?7, all alkalis have pH values?above?7
- The?lower?the pH then the?more acidic?the solution is
- The?higher?the pH then the?more alkaline?the solution is
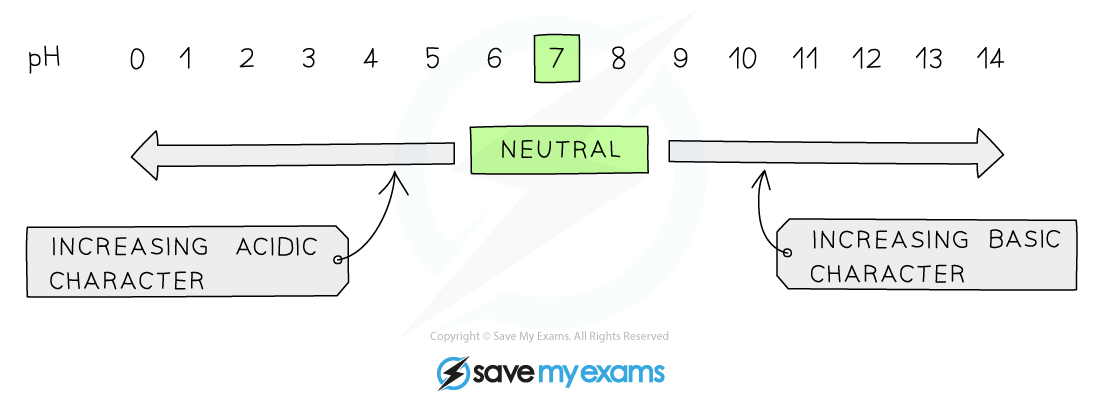
The pH scale showing acidity, neutrality and alkalinity
pH of water
- An equilibrium exists in water where few water molecules dissociate into proton and hydroxide ions
H2O(l) ? H+(aq) + OH-(aq)
- The equilibrium constant for this reaction is:
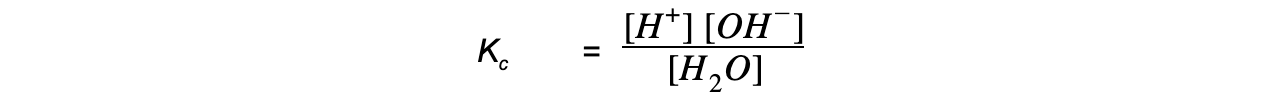
Kc?x [H2O] = [H+] [OH-]
- Since the concentration the H+?and OH-?ions is very small, the concentration of water is considered to be a constant, such that the expression can be rewritten as:
Kw?= [H+] [OH-]
Where?Kw?(ionic product of water)????? = ????????Kc?x [H2O]
=???????? 10-14?mol2?dm-3?at 298K
- Water at 298K has?equal amounts?of?OH-?and?H+?ions?with concentrations of?10-7?mol dm-3
- To calculate the pH of water, the following formula should be used:
 pH?? ??? = -log (10-7)
pH?? ??? = -log (10-7)
= 7
- Thus, water has a pH of 7
pH of acids
- Acidic?solutions (strong or weak)?always?have more H+?than OH-?ions
- Since the concentration of?H+?is always?greater?than the concentration of?OH-?ions, [H+] is always?greater?than 10-7?mol dm-3
- Using the pH formula, this means that the?pH of acidic solutions?is always?below?7
- The higher the [H+] of the acid, the lower the pH
pH of bases
- Basic?solutions (strong or weak)?always?have more?OH-than H+?ions
- Since the concentration of?OH-is always?greater?than the concentration of?H+?ions, [H+] is always?smaller?than 10-7?mol dm-3
- Using the pH formula, this means that the?pH of basic solutions?is always?above?7
- The higher the [OH-] of the base, the higher the pH
轉載自savemyexams
以上就是關于【CIE A Level Chemistry復習筆記1.7.10 pH Scale】的解答,如需了解學校/賽事/課程動態,可至翰林教育官網獲取更多信息。
往期文章閱讀推薦:
全網破防!ALevel CIE數學M1疑似錯題?經濟P2難度飆升?5月6日大考考情分析必看!
A-Level CIE就大規模泄題發布最嚴處罰!哪些考生必須重考?你的成績怎么辦?

最新發布
? 2026. All Rights Reserved. 滬ICP備2023009024號-1









