- 翰林提供學(xué)術(shù)活動(dòng)、國際課程、科研項(xiàng)目一站式留學(xué)背景提升服務(wù)!
- 021-63526630
CIE A Level Chemistry復(fù)習(xí)筆記1.7.9 Acid & Base Dissociation
Acid & Base Dissociation
Strong acids
- A?strong acid?is an acid that?dissociates?almost?completely?in aqueous solutions
- HCl (hydrochloric acid), HNO3?(nitric acid) and H2SO4?(sulfuric acid)
- The position of the equilibrium is so far over to the?right?that you can represent the reaction as an irreversible reaction
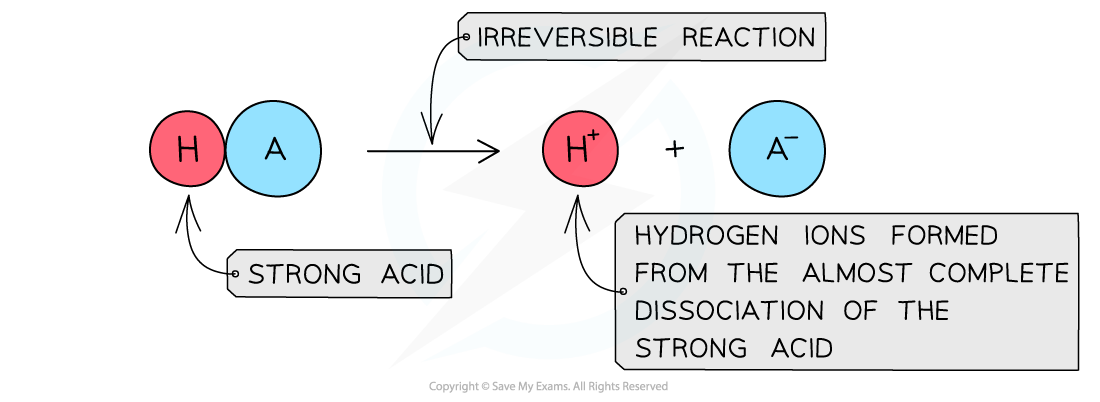
The diagram shows the almost complete dissociation of a strong acid in aqueous solution
- The solution formed is?highly acidic?due to the high concentration of the H+/H3O+?ions
- Since the?pH?depends on the concentration of H+/H3O+?ions, the pH can be calculated if the concentration of the strong acid is known
 pH is the negative log of the concentration of H+/H3O+?ions and can be calculated if the?concentration of the strong acid is known using the stoichiometry of the reaction
pH is the negative log of the concentration of H+/H3O+?ions and can be calculated if the?concentration of the strong acid is known using the stoichiometry of the reaction
Weak acids
- A?weak acid?is an acid that?partially?(or incompletely)?dissociates?in aqueous solutions
- Eg. most organic acids (ethanoic acid), HCN (hydrocyanic acid), H2S (hydrogen sulfide) and H2CO3?(carbonic acid)
- The position of the equilibrium is more over to the?left?and an equilibrium is established
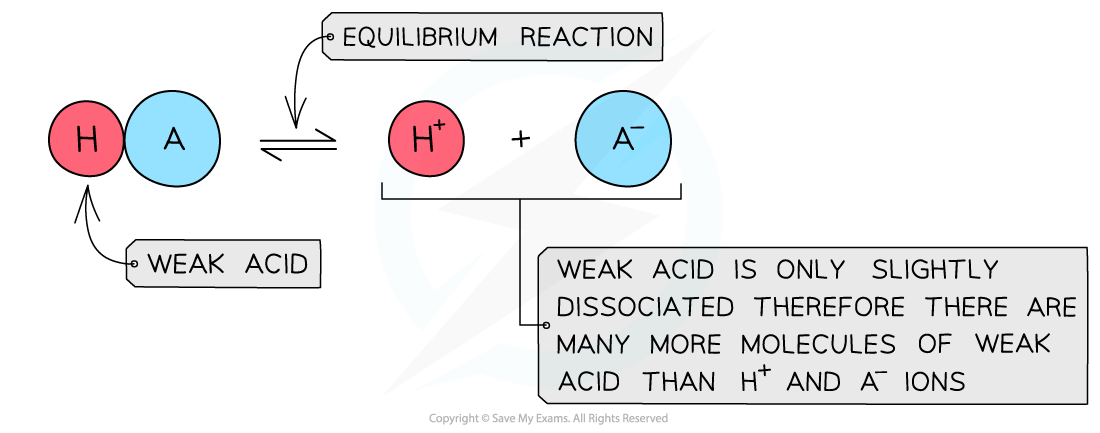
The diagram shows the almost complete dissociation of a weak acid in aqueous solution
- The solution is?less acidic?due to the lower concentration of H+/H3O+?ions
- Finding the?pH?of a weak acid is a bit more complicated as now the concentration of H+?ions is not equal to the concentration of acid
- To find the concentration of H+?ions, the acid dissociation constant (Ka) should be used
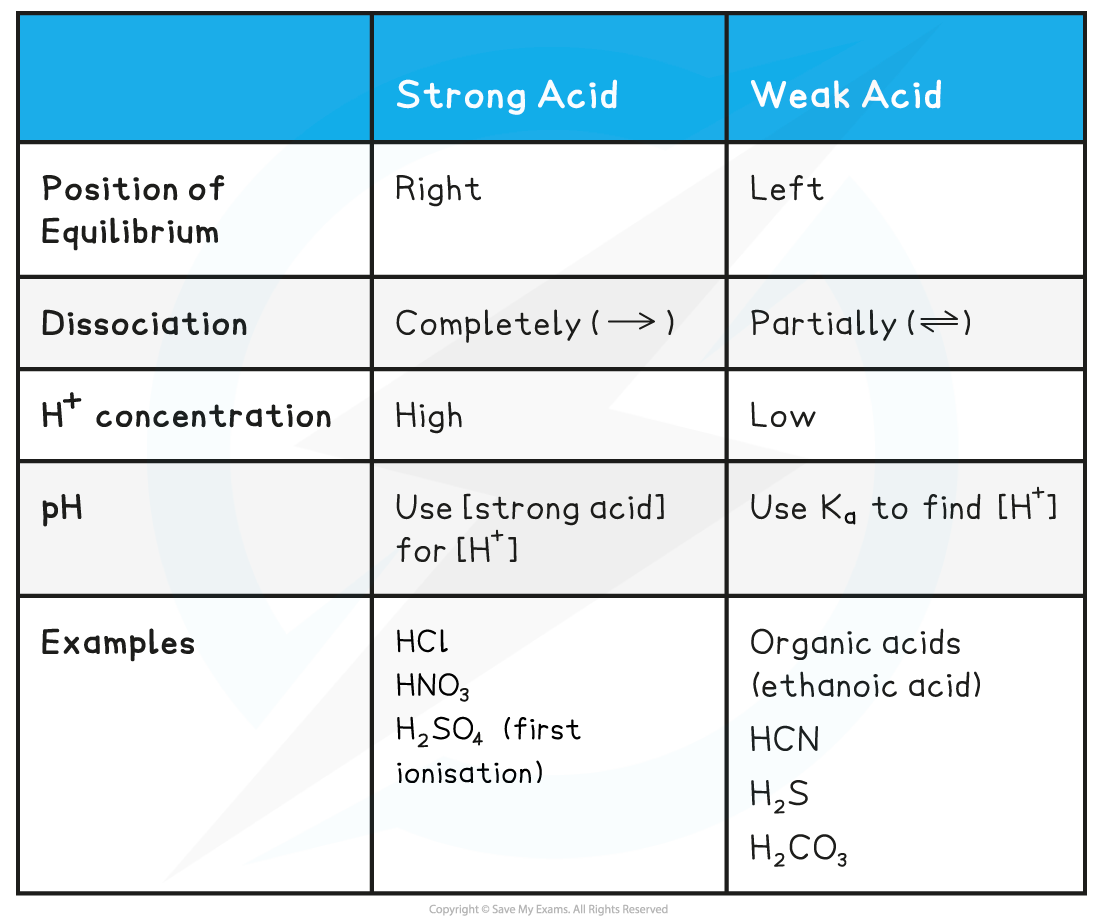
Acid & equilibrium position table
Strong bases
- A?strong base?is a base that dissociates almost completely in aqueous solutionsE.g. group 1 metal hydroxides such as NaOH (sodium hydroxide)
The position of the equilibrium is so far over to the right that you can represent the reaction as an irreversible reaction
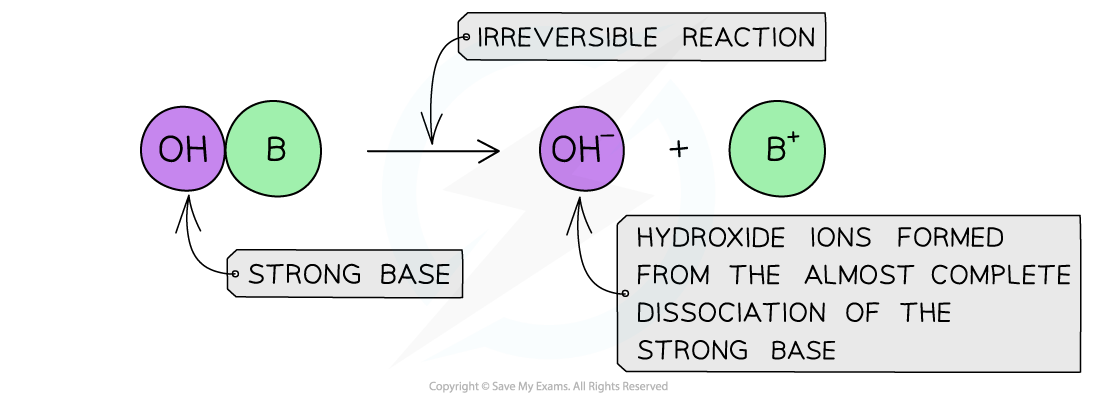 The diagram shows the almost complete dissociation of a strong base in aqueous solution
The diagram shows the almost complete dissociation of a strong base in aqueous solution
- The solution formed is highly basic due to the high concentration of the OH-?ions
Weak bases
- A?weak base?is a base that?partially?(or incompletely)?dissociates?in aqueous solutions
- NH3?(ammonia), amines and some hydroxides of transition metals
- The position of the equilibrium is more to the?left?and an equilibrium is established
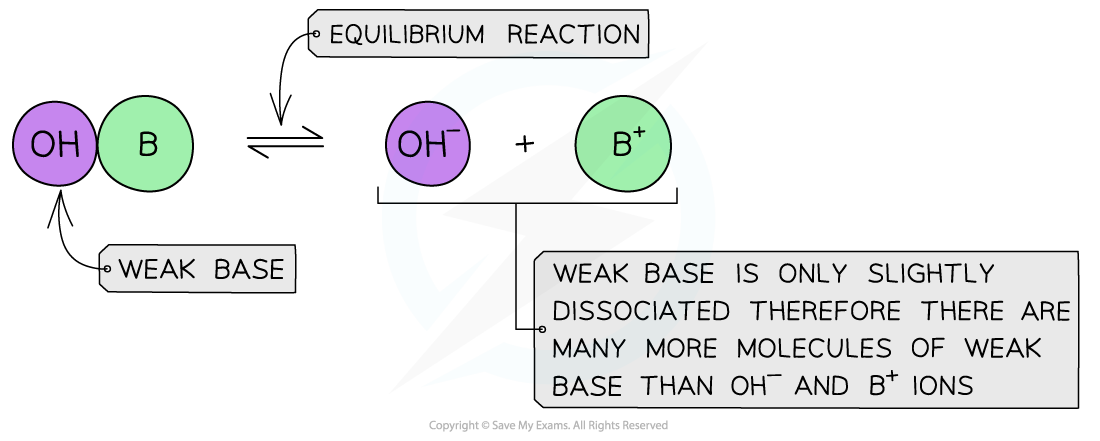
The diagram shows the almost complete dissociation of a weak base in aqueous solution
- The solution is?less basic?due to the lower concentration of OH-?ions
Base & equilibrium position table

Exam Tip
Hydrogen ions in aqueous solutions can be written as either as H3O+?or as H+?however, if H3O+?is used, H2O should be included in the chemical equation:HCl(g) → H+(aq) + Cl-(aq)orHCl(g) + H2O(l) → H3O+(aq) + Cl-(aq)?Remember that some acids are both strong and weak acids – for example,?H2SO4?(sulfuric acid) has two hydrogen ions that can ionise.H2SO4?acts as a strong acid:?H2SO4?→ H+?+ SO4-HSO4-?acts as a weak acid:?HSO4-? H+?+ SO42-?Also, don't forget that the terms?strong?and?weak?acids and bases are related to the?degree of dissociation?and not the?concentration.The appropriate terms to use when describing?concentration?are?dilute?and?concentrated.
轉(zhuǎn)載自savemyexams
以上就是關(guān)于【CIE A Level Chemistry復(fù)習(xí)筆記1.7.9 Acid & Base Dissociation】的解答,如需了解學(xué)校/賽事/課程動(dòng)態(tài),可至翰林教育官網(wǎng)獲取更多信息。
往期文章閱讀推薦:
全網(wǎng)破防!ALevel CIE數(shù)學(xué)M1疑似錯(cuò)題?經(jīng)濟(jì)P2難度飆升?5月6日大考考情分析必看!
A-Level CIE就大規(guī)模泄題發(fā)布最嚴(yán)處罰!哪些考生必須重考?你的成績?cè)趺崔k?

最新發(fā)布
? 2026. All Rights Reserved. 滬ICP備2023009024號(hào)-1









