- 翰林提供學術活動、國際課程、科研項目一站式留學背景提升服務!
- 021-63526630
CIE A Level Chemistry復習筆記1.7.3 Equilibrium Constant
Equilibrium Constant: Concentrations
Equilibrium expression & constant
- The?equilibrium expression?is an expression that links the?equilibrium constant,?Kc, to the?concentrations of reactants and products?at equilibrium taking the?stoichiometry?of the equation into account
- So, for a given reaction:
aA + bB ? cC + dD
the?Kc?is defined as follows:

Equilibrium expression linking the equilibrium concentration of reactants and products at equilibrium
- Solids?are ignored in equilibrium expressions
- The?Kc?of a reaction is specific and only changes if the?temperature?of the reaction changes
Worked example: Deducing equilibrium expressions

Answer

Mole Fraction & Partial Pressure
Partial pressure
- For reactions involving mixtures of gases, the equilibrium constant?Kp?is used as it is easier to measure the?pressure?than the concentration for gases
- The?partial pressure?of a gas is the pressure that the gas would have if it was in the container all by itself
- The?total pressure?is the sum of the?partial pressure

Equation to calculate the total pressure in a mixture of gases
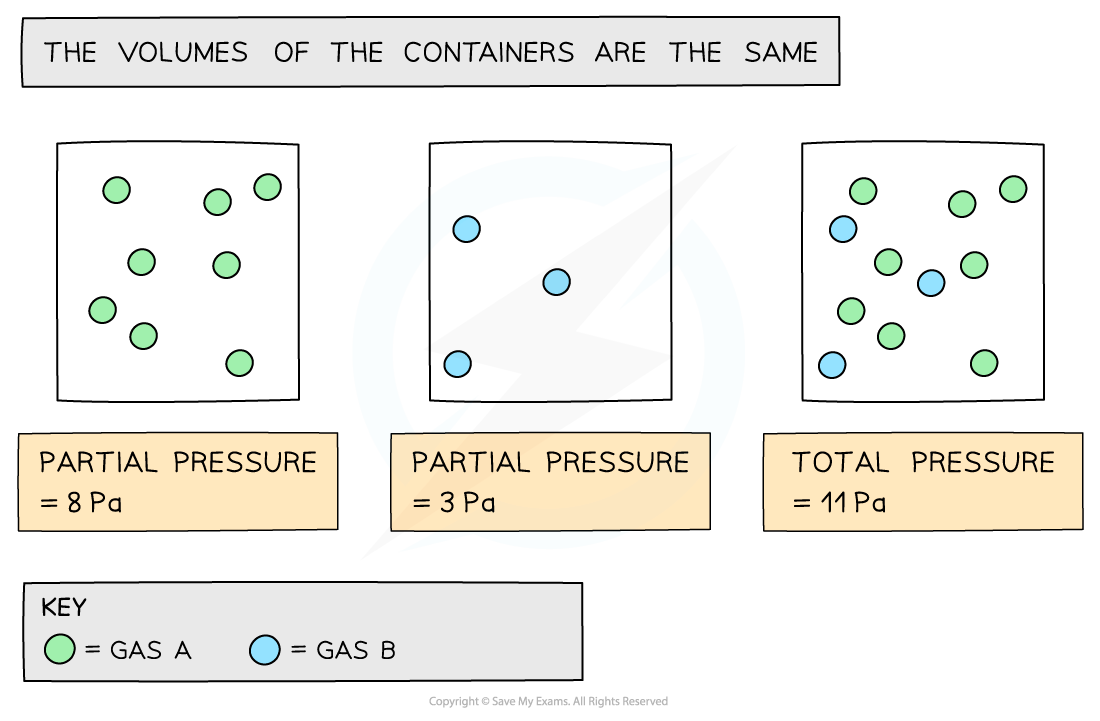 The diagram shows that each gas in the mixture has a partial pressure contributing towards the total pressure
The diagram shows that each gas in the mixture has a partial pressure contributing towards the total pressure
Mole fraction
- The mole fraction of a gas is the ratio of moles of a particular gas to the total number of moles of gas present
 Equation to calculate the mole fraction of a particular gas in a gas mixture
Equation to calculate the mole fraction of a particular gas in a gas mixture
- To calculate the?partial pressures of each gas?the following relationship can be used:
 Equation to calculate the partial pressure of a particular gas in a gas mixture
Equation to calculate the partial pressure of a particular gas in a gas mixture
The sum of the mole fractions should add up to 1.00, while the sum of the partial pressures should add up to the total pressure.
Equilibrium Constant: Partial Pressures
Equilibrium expressions involving partial pressures
- Equilibrium expressions in terms of partial pressures are written similarly to those involving concentrations with a few differences:
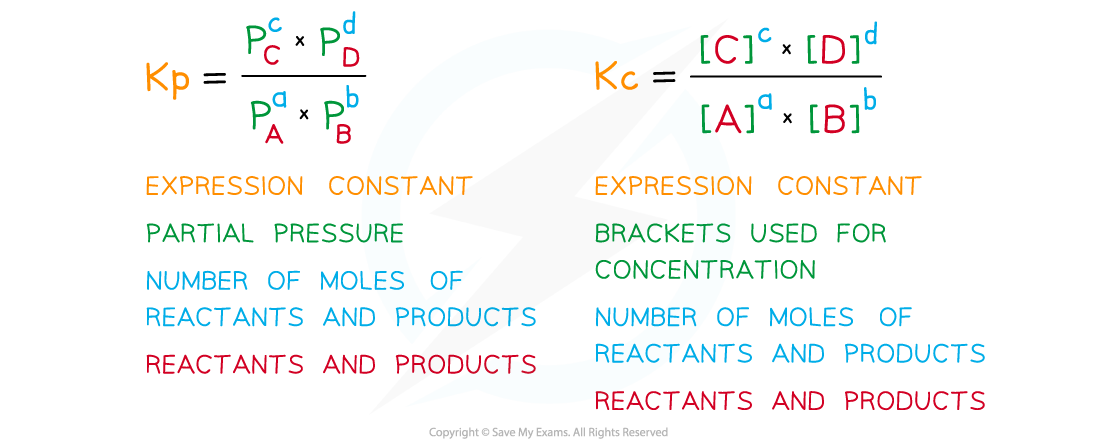 The diagram shows a comparison of writing equilibrium expressions in terms of partial pressures and concentrations
The diagram shows a comparison of writing equilibrium expressions in terms of partial pressures and concentrations
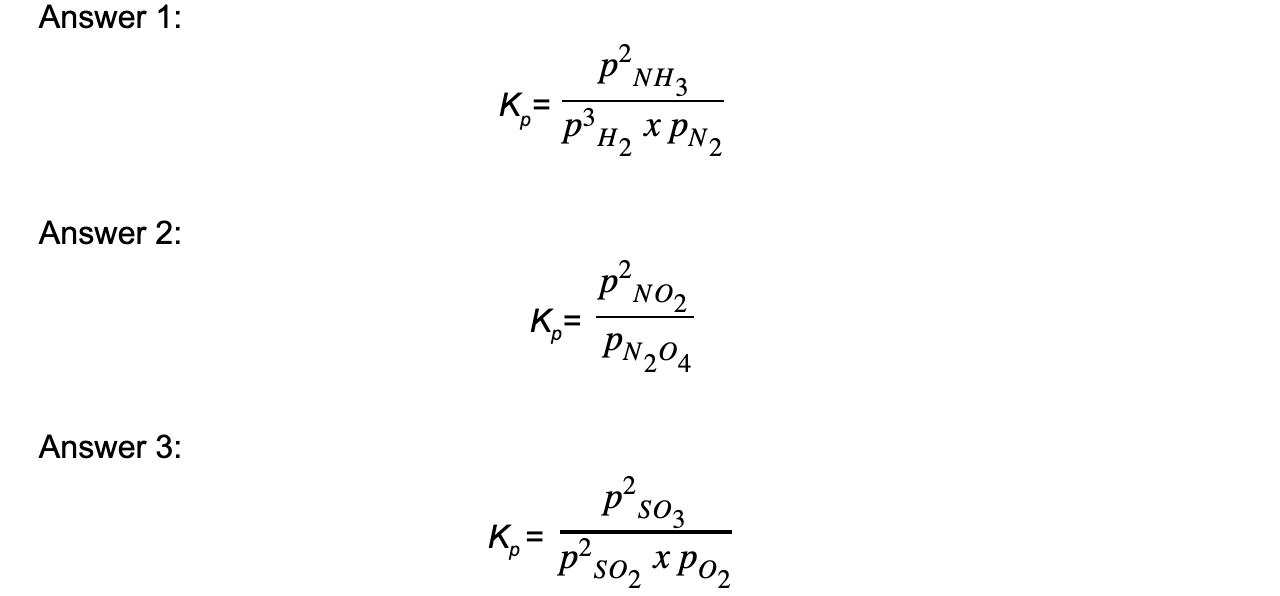
Worked example: Deducing equilibrium expressions of gaseous reactions
Answer
轉載自savemyexams
以上就是關于【CIE A Level Chemistry復習筆記1.7.3 Equilibrium Constant】的解答,如需了解學校/賽事/課程動態,可至翰林教育官網獲取更多信息。
往期文章閱讀推薦:
全網破防!ALevel CIE數學M1疑似錯題?經濟P2難度飆升?5月6日大考考情分析必看!
A-Level CIE就大規模泄題發布最嚴處罰!哪些考生必須重考?你的成績怎么辦?

最新發布
? 2026. All Rights Reserved. 滬ICP備2023009024號-1










