- 翰林提供學(xué)術(shù)活動(dòng)、國(guó)際課程、科研項(xiàng)目一站式留學(xué)背景提升服務(wù)!
- 021-63526630
CIE A Level Chemistry復(fù)習(xí)筆記1.7.4 Equilibrium Constant Calculations
Equilibrium Constant: Calculations
Calculations involving?Kc
- In the equilibrium expression each figure within a square bracket represents the concentration in?mol dm-3
- The?units?of?Kc?therefore depend on the form of the equilibrium expression
- Some questions give the?number of moles?of each of the reactants and products at equilibrium together with the volume of the reaction mixture
- The concentrations of the reactants and products can then be calculated from the number of moles and total volume

Equation to calculate concentration from number of moles and volume
Worked example: Calculating?Kc?of ethanoic acid

Answer
- Step 1:?Calculate the concentrations of the reactants and products
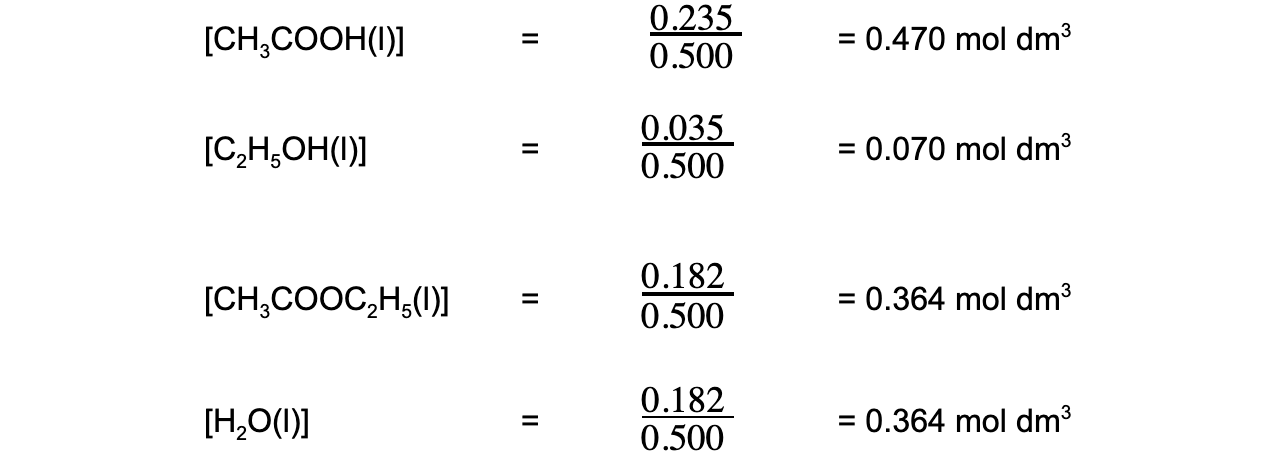
- Step 2:?Write out the balanced chemical equation with the concentrations of beneath each substance
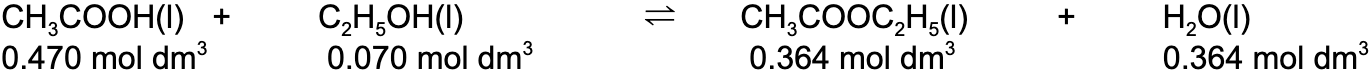
- Step 3:?Write the equilibrium constant for this reaction in terms of concentration
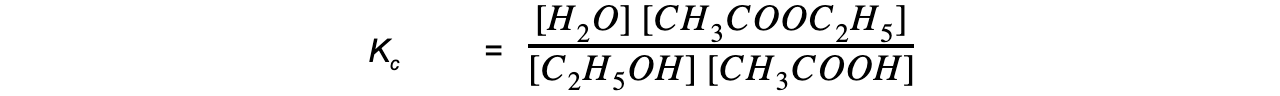
- Step 4:?Substitute the equilibrium concentrations into the expression
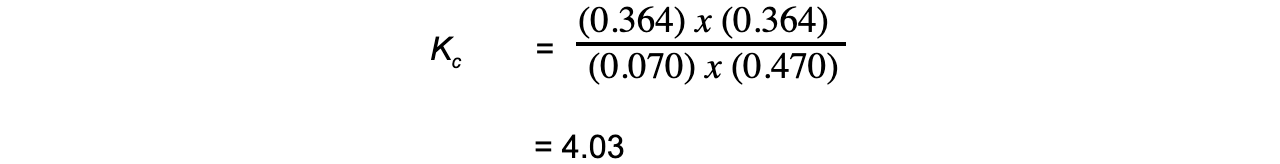
- Step 5:?Deduce the correct units for?Kc
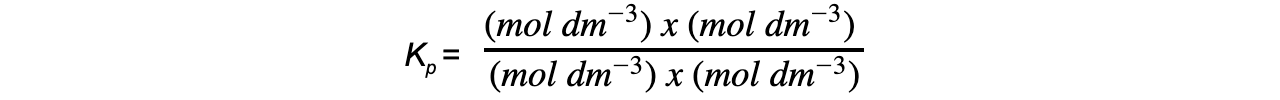
All units cancel out
Therefore,?Kc?= 4.03
- Note that the smallest number of significant figures used in the question is 3, so the final answer should also be given to 3 significant figures
- Some questions give the?initial and equilibrium concentrations?of the reactants but products
- An initial, change and equilibrium table should be used to determine the equilibrium concentration of the products?using the molar ratio of reactants and products in the stoichiometric equation
Worked example: Calculating Kc?of ethyl ethanoate
Answer
- Step 1:?Write out the balanced chemical equation with the concentrations of beneath each substance using an initial, change and equilibrium table
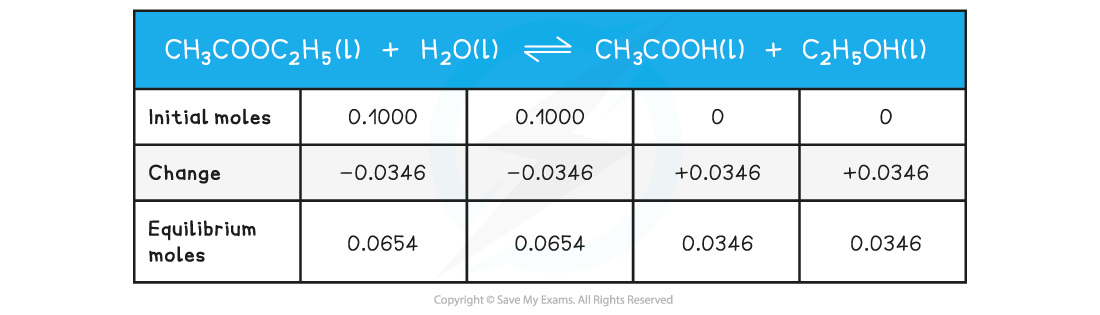
- Step 2:?Calculate the concentrations of the reactants and products
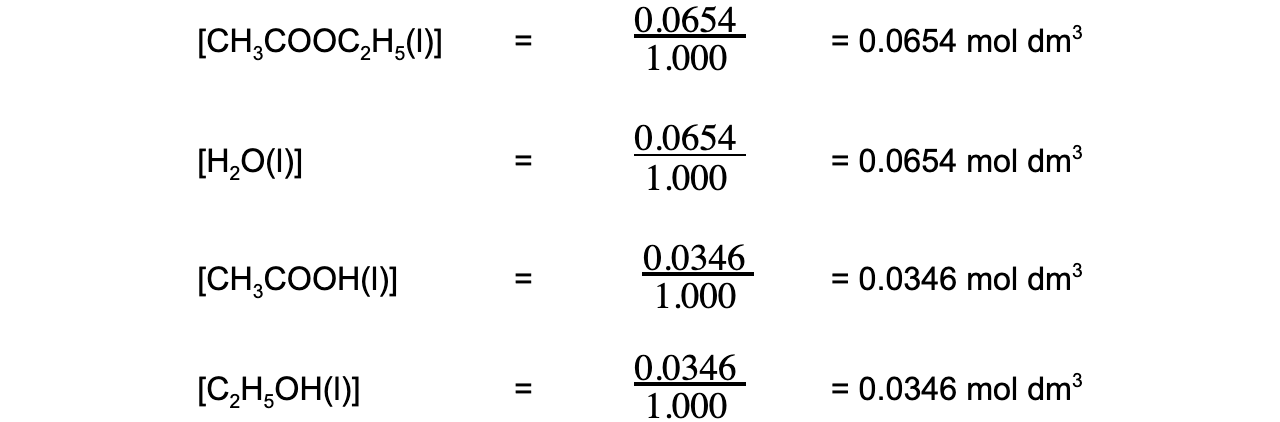
- Step 3:?Write the equilibrium constant for this reaction in terms of concentration
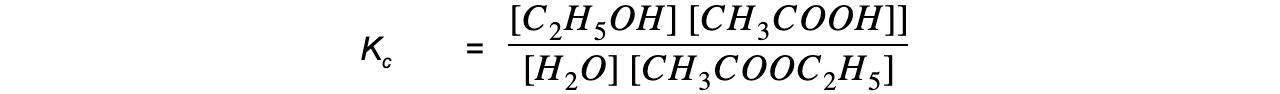
- Step 4:?Substitute the equilibrium concentrations into the expression
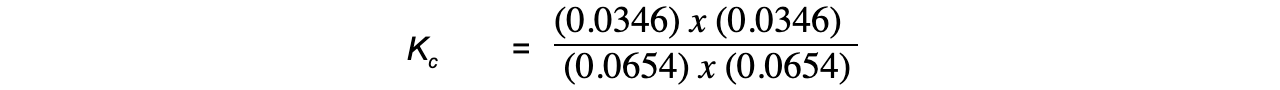
= 0.28
- Step 5:?Deduce the correct units for?Kc
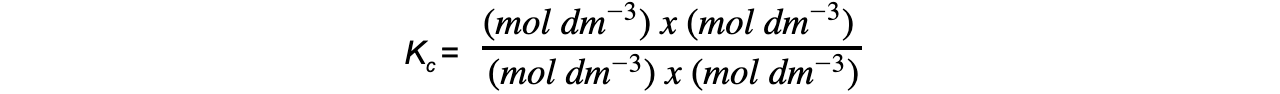
All units cancel out
Therefore, Kc = 0.288
Calculations involving?Kp
- In the equilibrium expression the?p?represent the partial pressure of the reactants and products in?Pa
- The?units?of?Kp?therefore depend on the form of the equilibrium expression
Worked example: Calculating?Kp?of a gaseous reaction

Answer
- Step 1:?Write the equilibrium constant for the reaction in terms of partial pressures
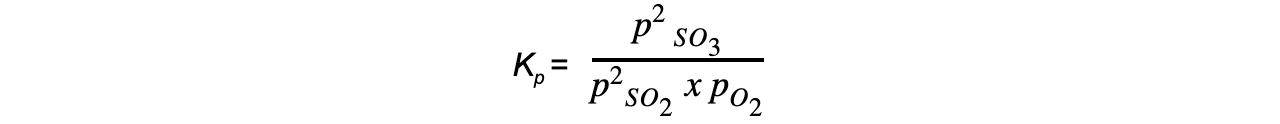
- Step 2:?Substitute the equilibrium concentrations into the expression
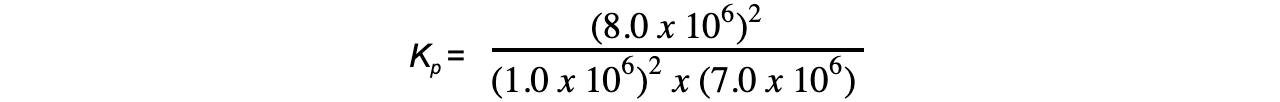
= 9.1 x 10-6
- Step 3:?Deduce the correct units of?Kp
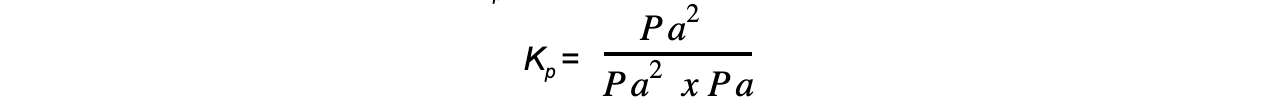
The units of?Kp?are Pa-1
Therefore, Kp = 9.1 x 10-6 Pa-1
- Some questions only give the?number of moles?of gases present and the total pressure
- The number of moles of each gas should be used to first calculate the?mole fractions
- The mole fractions are then used to calculate the?partial pressures
- The values of the partial pressures are then substituted in the?equilibrium expression
Worked example: Calculating?Kp?of hydrogen iodide equilibrium reaction

- Step 1:?Calculate the total number of moles
Total number of moles = 1.71 x 10-3?+ 2.91 x 10-3?+ 1.65 x 10-2
= 2.112 x 10-2
- Step 2:?Calculate the mole fraction of each gas
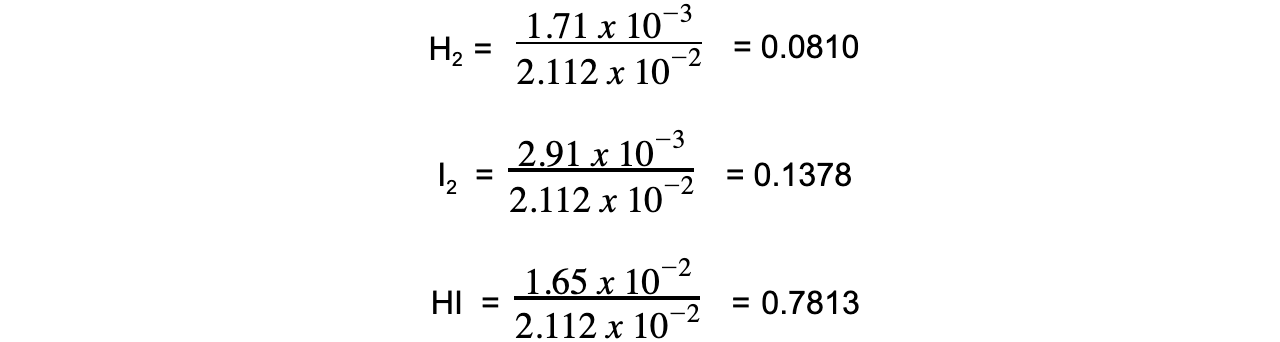
- Step 3:?Calculate the partial pressure of each gas
H2?= 0.0810 x 100 = 8.10 kPa
I2?= 0.1378 x 100 = 13.78 kPa
HI = 0.7813 x 100 = 78.13 kPa
- Step 4:?Write the equilibrium constant in terms of partial pressure
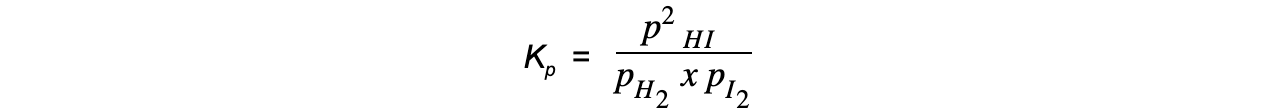
- Step 5:?Substitute the values into the equilibrium expression
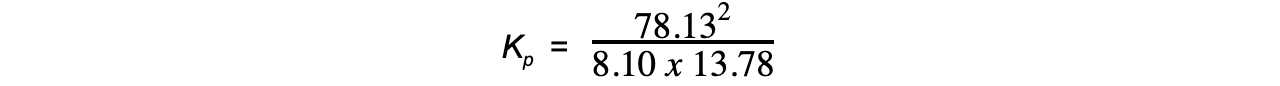
= 54.7
- Step 6:?Deduce the correct units for?Kp
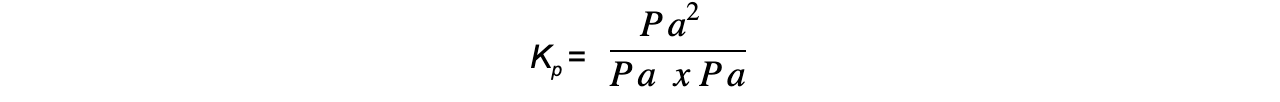
All units cancel out
Therefore, Kp = 54.7
- Other questions related to equilibrium expressions may involve calculating quantities present at equilibrium given appropriate data
Worked example: Calculating partial pressures

Answer
There are equal volumes of reactants A and B in a 1:1 molar ratio.
This means their partial pressures will be the same.B therefore also has an equilibrium partial pressure of 0.5
Total pressure = sum of equilibrium (Σ) partial pressures
Therefore, the sum of all the partial pressures must equal to 3 atm
0.5 + 0.5 +?pc?= 3 atm
pc?= 2 atm
轉(zhuǎn)載自savemyexams
以上就是關(guān)于【CIE A Level Chemistry復(fù)習(xí)筆記1.7.4 Equilibrium Constant Calculations】的解答,如需了解學(xué)校/賽事/課程動(dòng)態(tài),可至翰林教育官網(wǎng)獲取更多信息。
往期文章閱讀推薦:
全網(wǎng)破防!ALevel CIE數(shù)學(xué)M1疑似錯(cuò)題?經(jīng)濟(jì)P2難度飆升?5月6日大考考情分析必看!
A-Level CIE就大規(guī)模泄題發(fā)布最嚴(yán)處罰!哪些考生必須重考?你的成績(jī)?cè)趺崔k?

最新發(fā)布
? 2026. All Rights Reserved. 滬ICP備2023009024號(hào)-1










