- 翰林提供學術活動、國際課程、科研項目一站式留學背景提升服務!
- 021-63526630
CIE A Level Chemistry復習筆記1.3.12 Hydrogen Bonding
Hydrogen Bonding
Hydrogen bonding
- Hydrogen bonding?is the?strongest?form of?intermolecular bonding
- Intermolecular bonds are bonds?between?molecules
- Hydrogen bonding is a type of?permanent dipole – permanent dipole?bonding
- For hydrogen bonding to take place the following is needed:
- A species which has an?O?or?N?(very?electronegative) atom with an available?lone pair?of electrons
- A species with an?-OH?or?-NH?group
- When hydrogen is covalently bonded to an?electronegative?atom, such as?O?or?N, the bond becomes very highly?polarised
- The H becomes so?δ+?charged that it can form a bond with the?lone pair?of an?O?or?N atom?in another molecule
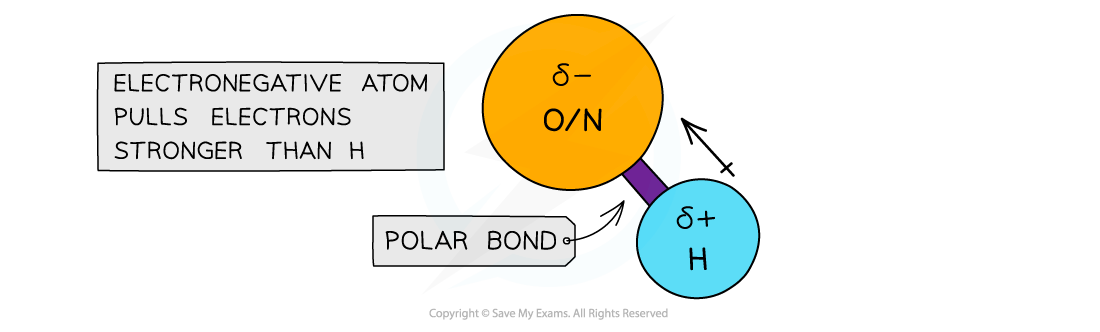
The electronegative atoms O or N have a stronger pull on the electrons in the covalent bond with hydrogen, causing the bond to become polarised
- For hydrogen bonding to take place, the?angle?between the?-OH/-NH?and the?hydrogen bond?is?180o
- The number of hydrogen bonds depends on:
- The number of hydrogen atoms attached to O or N in the molecule
- The number of?lone?pairs?on the O or N
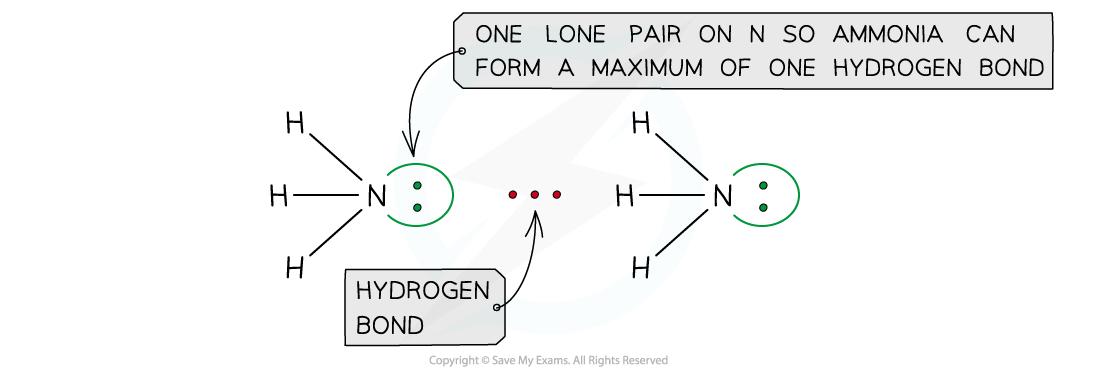
Ammonia can form a maximum of one hydrogen bond per molecule
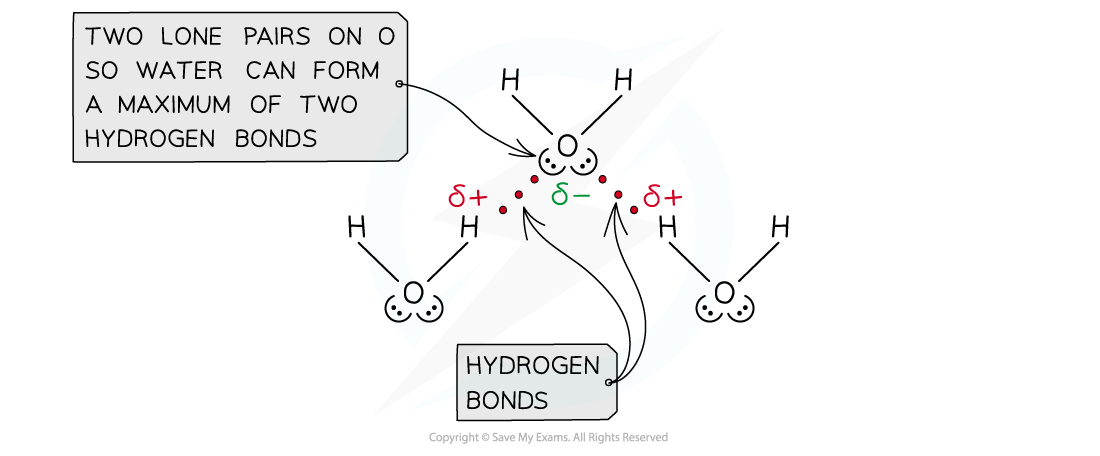
Water can form a maximum of two hydrogen bonds per molecule
Properties of water
- Hydrogen bonding in water, causes it to have?anomalous properties?such as high melting and boiling points, high surface tension and anomalous density of ice compared to water
High melting & boiling points
- Water has high?melting?and?boiling points?which is caused by the?strong intermolecular forces?of hydrogen bonding between the molecules
- In?ice?(solid H2O) and water (liquid H2O) the molecules are tightly held together by hydrogen bonds
- A lot of energy is therefore required to break the water molecules apart and melt or boil them
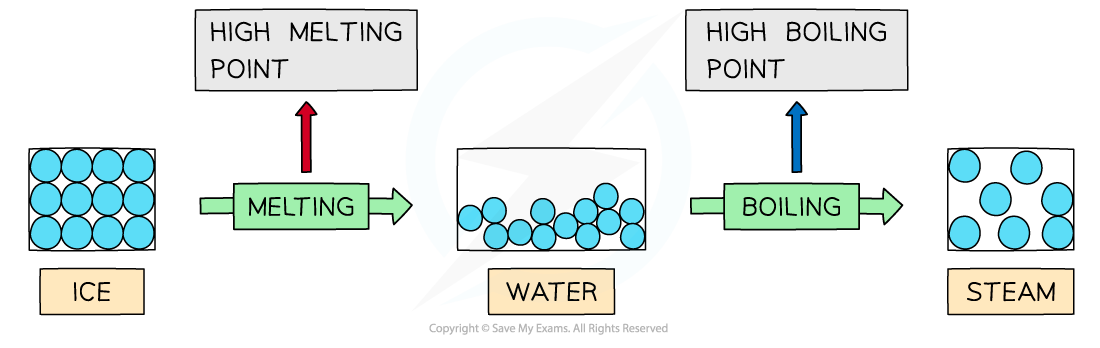 Hydrogen bonds are strong intermolecular forces which are difficult to break causing water to have high melting and boiling points
Hydrogen bonds are strong intermolecular forces which are difficult to break causing water to have high melting and boiling points
- The graph below compares the?enthalpy of vaporisation?(energy required to boil a substance) of different hydrides
- The enthalpy changes?increase?going from H2S to H2Te due to the increased number of electrons in the Group 16 elements
- This causes an?increased instantaneous dipole - induced dipole forces?as the molecules become larger
- Based on this, H2O would have a much lower enthalpy change (around 17 kJ mol-1)
- However, the enthalpy change of vaporisation is almost 3 times?larger?which is caused by the?hydrogen bonds?present in water but not in the other hydrides
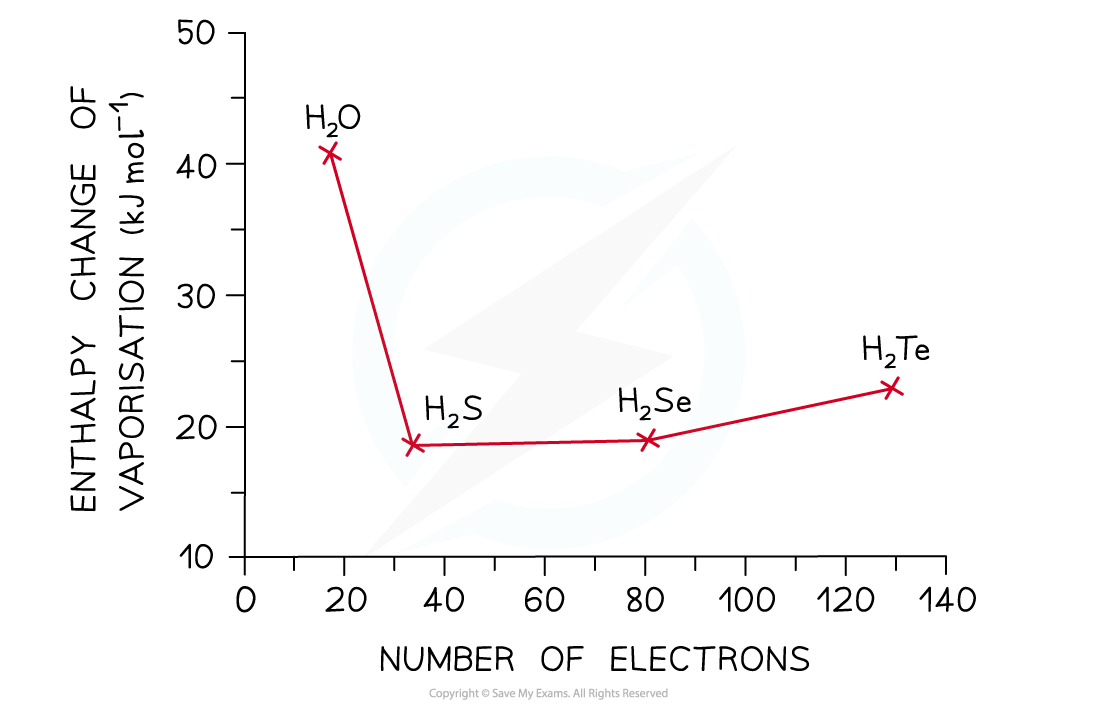
The high enthalpy change of evaporation of water suggests that instantaneous dipole-induced dipole forces are not the only forces present in the molecule – there are also those of the strong hydrogen bonds, which cause the high boiling points
High surface tension
- Water has a?high surface tension
- Surface?tension?is the ability of a?liquid surface?to resist any?external forces?(i.e. to stay unaffected by forces acting on the surface)
- The water molecules at the?surface?of liquid are bonded to other water molecules through?hydrogen?bonds
- These molecules?pull downwards?on the?surface molecules?causing the surface them to become compressed and more tightly together at the surface
- This increases water’s?surface?tension
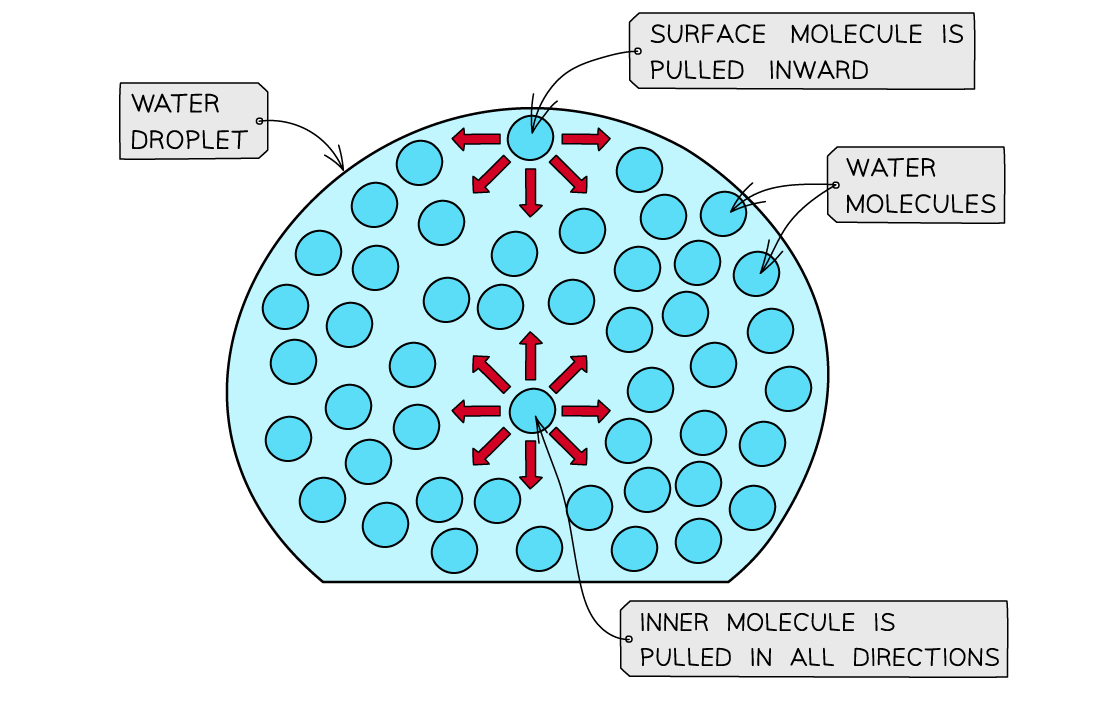
The surface molecules are pulled downwards due to the hydrogen bonds with other molecules, whereas the inner water molecules are pulled in all directions
Density
- Solids?are?denser?than their?liquids?as the particles in solids are more?closely packed?together than in their liquid state
- In ice however, the water molecules are packed in a?3D?hydrogen-bonded?network in a?rigid lattice
- Each oxygen atom is surrounded by hydrogen atoms
- This way of packing the molecules in a solid and the relatively long?bond lengths?of the hydrogen bonds means that the water molecules are slightly further apart than in the liquid form
- Therefore, ice has a lower density than liquid water
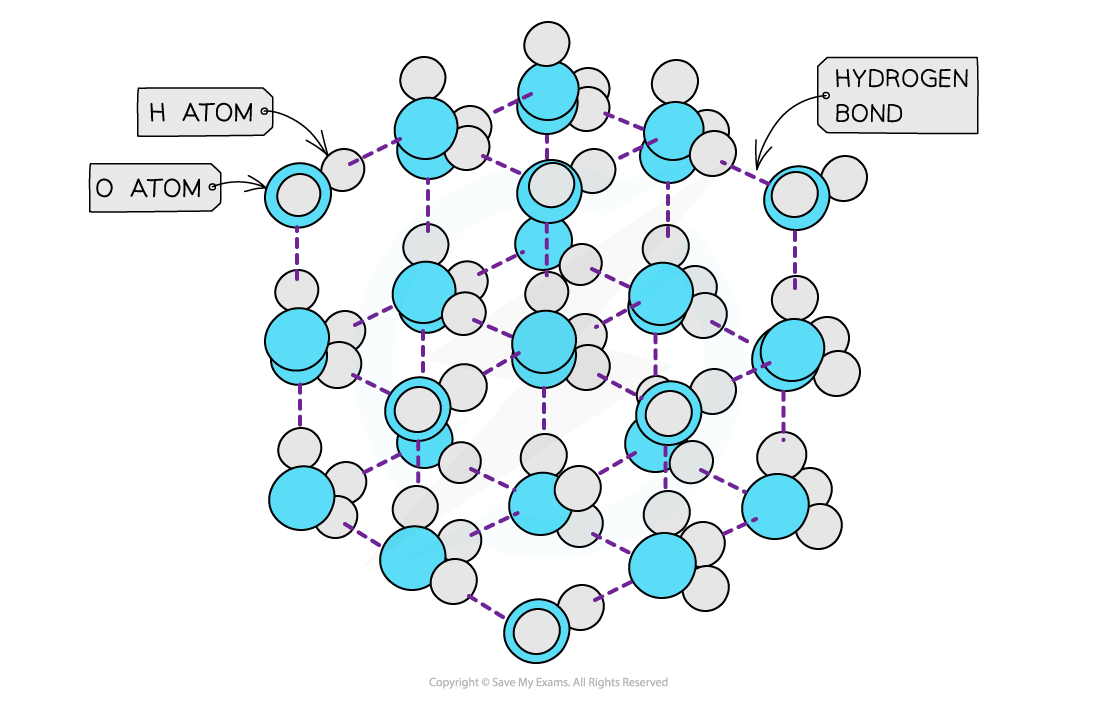
The ‘more open’ structure of molecules in ice causes it to have a lower density than liquid water
Exam Tip
Ice floats on water because of ice's lower density.
轉載自savemyexams
以上就是關于【CIE A Level Chemistry復習筆記1.3.12 Hydrogen Bonding】的解答,如需了解學校/賽事/課程動態,可至翰林教育官網獲取更多信息。
往期文章閱讀推薦:
全網破防!ALevel CIE數學M1疑似錯題?經濟P2難度飆升?5月6日大考考情分析必看!
A-Level CIE就大規模泄題發布最嚴處罰!哪些考生必須重考?你的成績怎么辦?

最新發布
? 2026. All Rights Reserved. 滬ICP備2023009024號-1









