- 翰林提供學術活動、國際課程、科研項目一站式留學背景提升服務!
- 021-63526630
CIE A Level Chemistry復習筆記1.3.10 Bond Energy & Length
Covalent Bonding: Bond Energy & Length
Bond energy
- The?bond energy?is the energy required to?break?one mole of a particular covalent bond in the gaseous states
- Bond energy has units of kJ mol-1
- The?larger?the bond energy, the?stronger?the covalent bond is
Bond length
- The?bond length?is?internuclear distance of two covalently bonded atoms
- It is the distance from the nucleus of one atom to another atom which forms the covalent bond
- The?greater?the forces of attraction between electrons and nuclei, the more the atoms are pulled closer to each other
- This?decreases?the?bond?length?of a molecule and?increases?the?strength?of the covalent bond
- Triple bonds?are the?shortest?and?strongest?covalent bonds due to the large electron density between the nuclei of the two atoms
- This increase the forces of attraction between the electrons and nuclei of the atoms
- As a result of this, the atoms are pulled closer together causing a shorter bond length
- The increased forces of attraction also means that the covalent bond is?stronger

Triple bonds are the shortest covalent bonds and therefore the strongest ones
Reactivity of covalent molecules
- The?reactivity?of a covalent bond is greatly influenced by:
- The bond?polarity
- The bond?strength
- The bond?type?(σ/π)
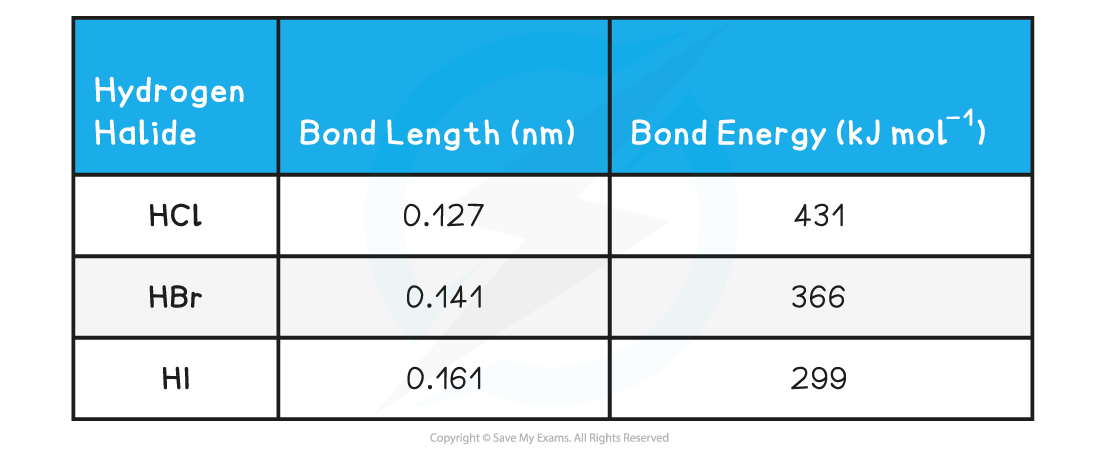
Worked example: Bond lengths & bond energies

Answer
Answer 1:?Going down the halogen group, the atoms are bigger; the attractive force between the bonding electrons and the nucleus get smaller so less energy is needed to break the atom
Answer 2:?Going down the group the increase in bond length is approximately 0.14-0.20 nm
Fluorine is smaller than HCl, so a value between 0.09 and 0.11 nm is acceptable for the bond length
Answer 3:?The hydrogen halide with the longest bond length and therefore smallest bond energy is the most reactive as it takes the least energy to break apart the hydrogen and halide atoms apart
Therefore, HI is the most reactive hydrogen halide
轉載自savemyexams
以上就是關于【CIE A Level Chemistry復習筆記1.3.10 Bond Energy & Length】的解答,如需了解學校/賽事/課程動態,可至翰林教育官網獲取更多信息。
往期文章閱讀推薦:
全網破防!ALevel CIE數學M1疑似錯題?經濟P2難度飆升?5月6日大考考情分析必看!
A-Level CIE就大規模泄題發布最嚴處罰!哪些考生必須重考?你的成績怎么辦?

最新發布
? 2026. All Rights Reserved. 滬ICP備2023009024號-1









