- 翰林提供學(xué)術(shù)活動(dòng)、國際課程、科研項(xiàng)目一站式留學(xué)背景提升服務(wù)!
- 021-63526630
CIE A Level Chemistry復(fù)習(xí)筆記1.7.7 Acids & Bases
Common Acids
- An?acid?is a substance that?neutralises?a base forming a?salt?and?water:
2HCl(aq) + CaO(s) ? CaCl2(aq) + H2O(l)
acid??????????????????????????????? salt

Acids react with bases to form salt and water
- Acids are also substances that release?hydrogen ions?when they dissolve in water:
HCl(g) → H+(aq) + Cl-?(aq)
 Acids dissociate in water to release a hydrogen ion
Acids dissociate in water to release a hydrogen ion
Names & formulae of some common acids table
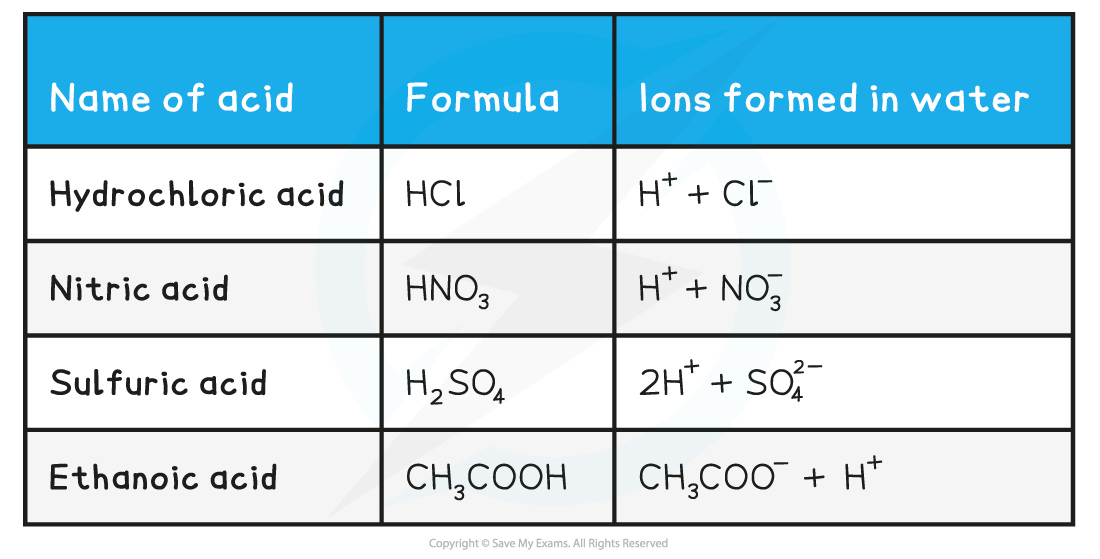
- In organic acids (such as carboxylic acids) only some of the hydrogen atoms can form ions when the acid dissociates
Common Alkalis
- A?base?is a compound that?neutralises?an acid forming a?salt?and?water
2HCl(aq) + CaO(s) ? CaCl2(aq) + H2O(l)
base? ? ? ? ? salt
- A base is a substance that?accepts?hydrogen ions or a compound that contains?oxide?or?hydroxide?ions
- For example:
NH3(g) + H2O(l) → NH4+(aq) + OH-(aq)
base? ? ? ? ? ? ? ? ? ? ? ? ? ? ? ? hydroxide ions
For example:
NaOH(s) + aq → Na+(aq) + OH-(aq)
base? ? ? ? ? ? ? ? ? ? ? ? ? ? ? ? hydroxide ions
- A base that is?soluble?in water is called an?alkali
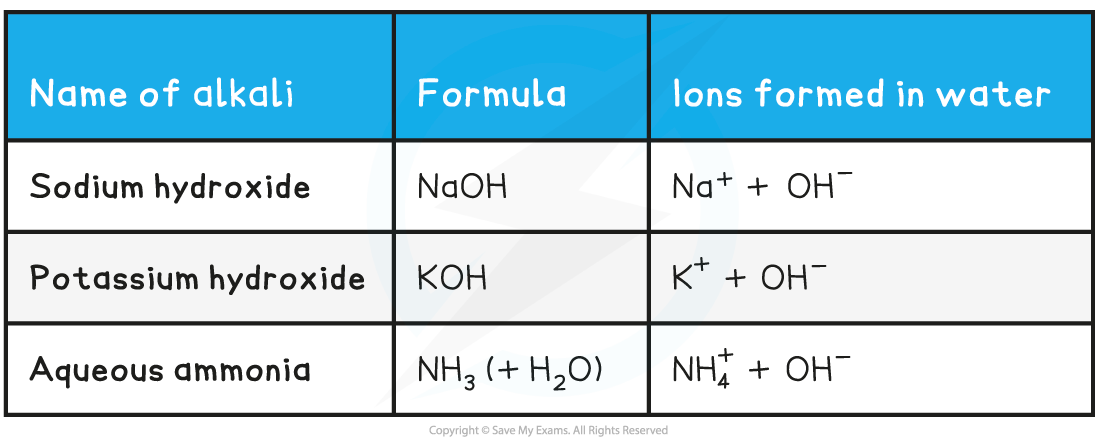
Names & formulae of some common alkalis table
轉(zhuǎn)載自savemyexams
以上就是關(guān)于【CIE A Level Chemistry復(fù)習(xí)筆記1.7.7 Acids & Bases】的解答,如需了解學(xué)校/賽事/課程動(dòng)態(tài),可至翰林教育官網(wǎng)獲取更多信息。
往期文章閱讀推薦:
全網(wǎng)破防!ALevel CIE數(shù)學(xué)M1疑似錯(cuò)題?經(jīng)濟(jì)P2難度飆升?5月6日大考考情分析必看!
A-Level CIE就大規(guī)模泄題發(fā)布最嚴(yán)處罰!哪些考生必須重考?你的成績?cè)趺崔k?

最新發(fā)布
? 2026. All Rights Reserved. 滬ICP備2023009024號(hào)-1









