- 翰林提供學(xué)術(shù)活動、國際課程、科研項(xiàng)目一站式留學(xué)背景提升服務(wù)!
- 021-63526630
Edexcel IGCSE Chemistry 復(fù)習(xí)筆記 2.2.1 Group 7 (Halogens)
Edexcel IGCSE Chemistry 復(fù)習(xí)筆記 2.2.1 Group 7 (Halogens)
Physical Properties
- The elements in group 7 are known as the halogens
- These are fluorine, chlorine, bromine, iodine and astatine
- These elements are non-metals that are?poisonous
- All halogens have similar reactions as they each have seven electrons in their outermost shell
- Halogens are?diatomic, meaning they form molecules made of pairs of atoms sharing electrons (forming a single covalent bond between the two halogen atoms)
Trends in Physical Properties
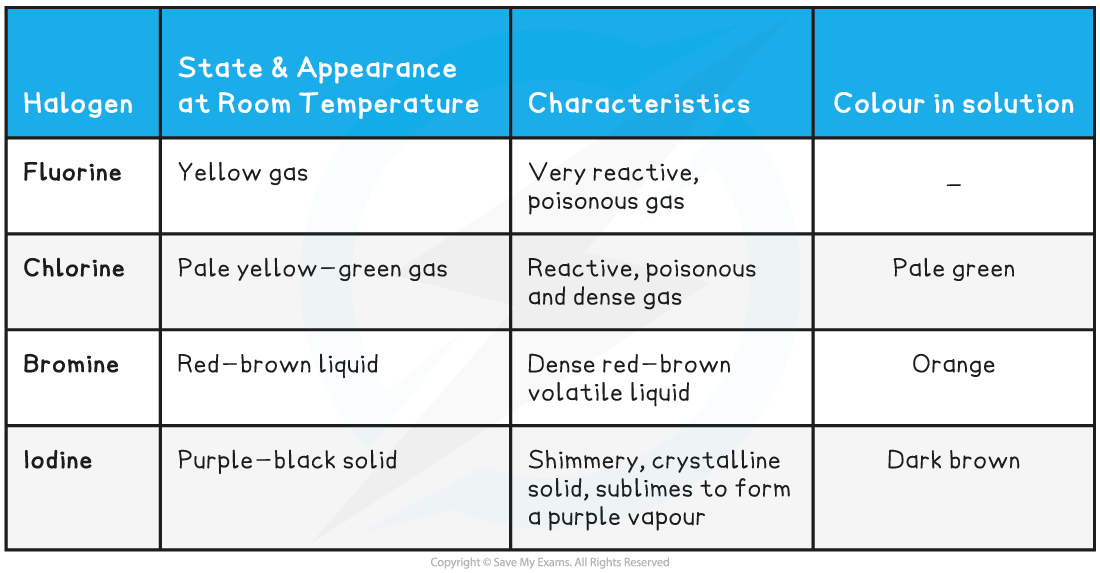
- At room temperature, the halogens exist in different states and colours, with different characteristics
The Appearance, Characteristics and Colour in Solution of the Halogens
- The melting and boiling points of the halogens?increase?as you go down the group
- This is due to increasing intermolecular forces as the atoms become larger, so more energy is required to overcome these forces
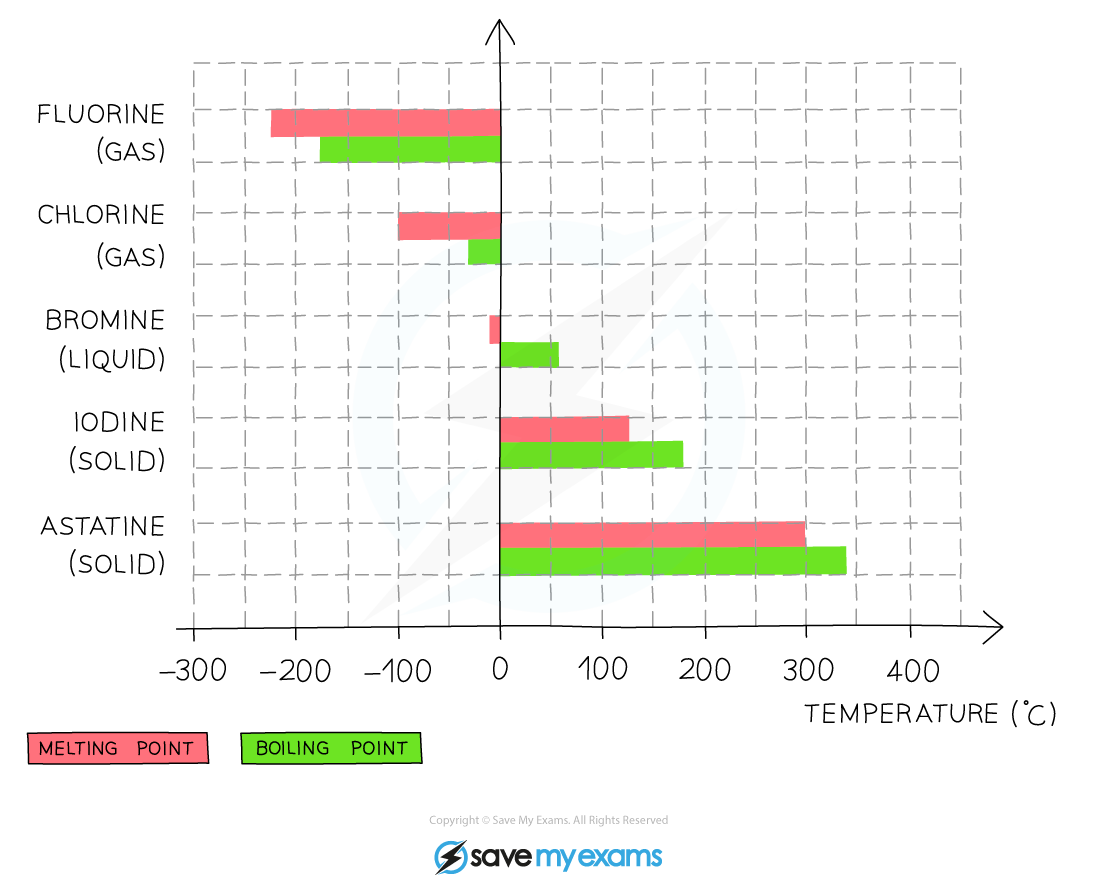
This graph shows the melting and boiling points of the group 7 halogens?
- At room temperature (20 °C), the physical state of the halogens changes as you go down the group
- Fluorine and chlorine are?gases, bromine is a?liquid?and iodine is crumbly?solid
- The colours of the halogens also change as you descend the group - they become darker

The physical states and colours of chlorine, bromine and iodine at room temperature?
Exam Tip
Exam questions on this topic occur often so make sure you know and can explain the trends of the group 7 elements in detail, using their electron configurations.
Predicting Properties in Group 7
- Chlorine, bromine and iodine react with metals and non-metals to form compounds
Metal Halides
- The halogens react with some metals to form?ionic compounds?which are?metal?halide?salts
- The halide ion carries a -1 charge so the ionic compound formed will have different numbers of halogen atoms, depending on the?valency?of the metal
- E.g., sodium is a group 1 metal:
- 2 Na + Cl2?→ 2 NaCl
- Calcium is a group 2 metal:
- Ca + Br2?→ CaBr2
- The halogens?decrease?in?reactivity?moving down the group, but they still form halide salts with some metals including iron
- The rate of reaction is?slower?for halogens which are?further?down the group such as bromine and iodine
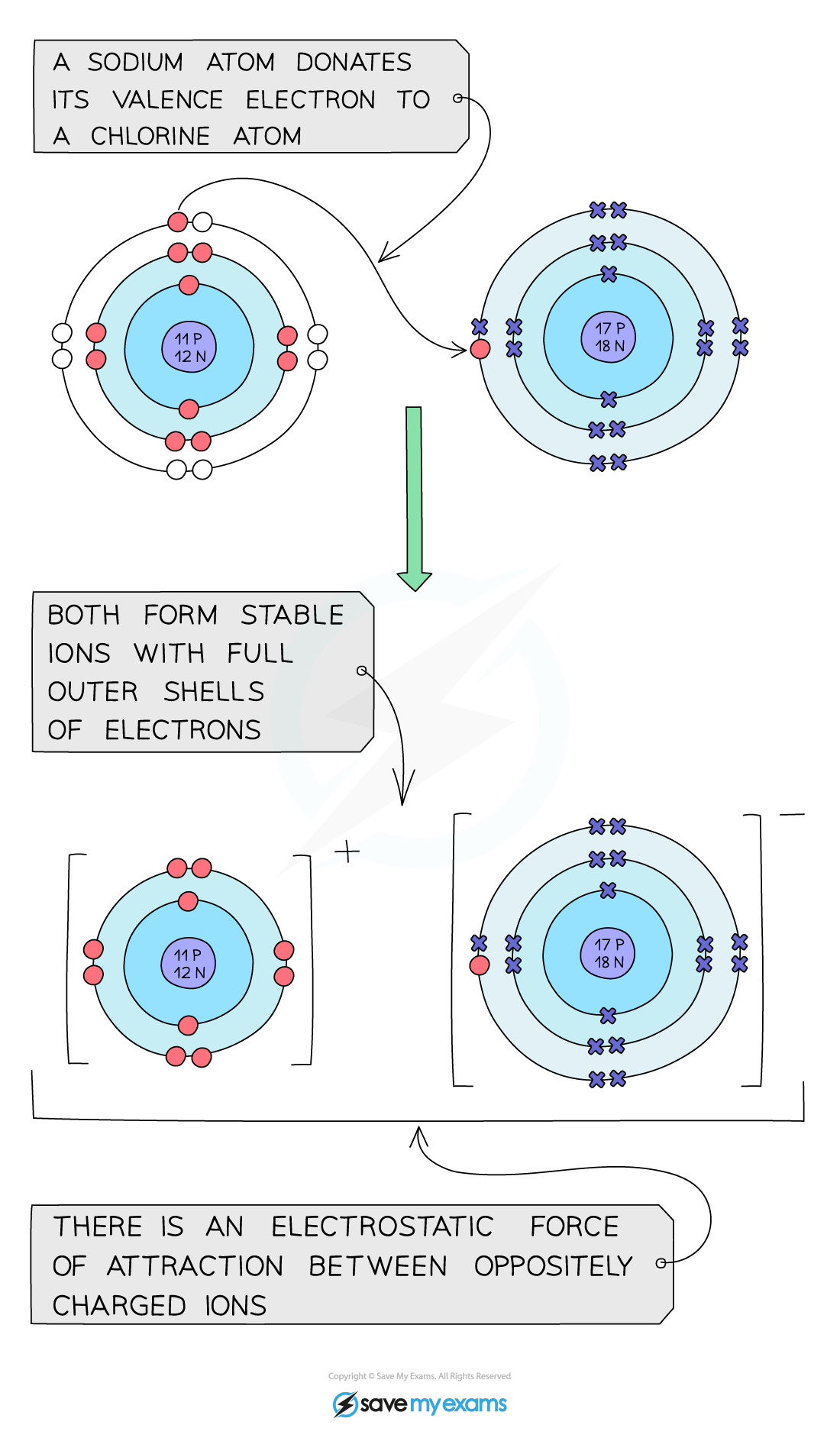
Sodium donates its single outer electron to a chlorine atom and an ionic bond is formed between the positive sodium ion and the negative chloride ion
Non-metal Halides
- The halogens react with non-metals to form simple molecular covalent structures
- For example, the halogens react with hydrogen to form?hydrogen halides?(e.g., hydrogen chloride)
- Reactivity decreases down the group, so iodine reacts less vigorously with hydrogen than chlorine (which requires light or a high temperature to react with hydrogen)
- Fluorine is the most reactive (reacting with hydrogen at low temperatures in the absence of light)
Displacement Reactions
- A?halogen displacement reaction?occurs when a more reactive halogen displaces a less reactive halogen from an aqueous solution of its halide
- The reactivity of group 7 elements decreases as you move down the group
- You only need to learn the displacement reactions with chlorine, bromine and iodine
- Chlorine is the most reactive and iodine is the least reactive
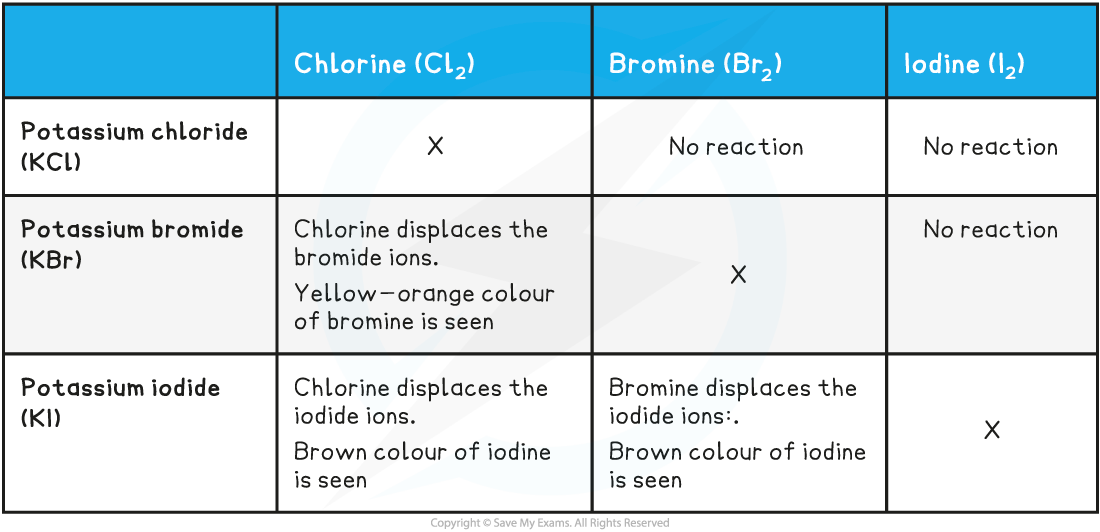
Chlorine with Bromides & Iodides
- If you add chlorine solution to colourless potassium bromide or potassium iodide solution a displacement reaction occurs:
- The solution becomes orange as bromine is formed or
- The solution becomes brown as iodine is formed
- Chlorine is?above?bromine and iodine in group 7 so it is more reactive
- Chlorine will?displace?bromine or iodine from an aqueous solution of the metal halide:
Cl2?+ 2KBr?→?2KCl?+ Br2
chlorine + potassium bromide?→??potassium chloride + bromine
Cl2?+ 2KI?→?2KCl?+ I2
chlorine + potassium iodide?→??potassium chloride + iodine
Bromine with Iodides
- Bromine is above iodine in group 7 so it is?more?reactive
- Bromine will displace iodine from an aqueous solution of the metal iodide
bromine + potassium iodide?→??potassium bromide + iodine
Br2?+ 2KI → 2KBr + I2
- This table shows a summary of the displacement reactions of the halogens: chlorine, bromine and iodine?
Exam Tip
Displacement reactions are sometimes known as single replacement reactions.
轉(zhuǎn)載自savemyexam
以上就是關(guān)于【Edexcel IGCSE Chemistry 復(fù)習(xí)筆記 2.2.1 Group 7 (Halogens)】的解答,如需了解學(xué)校/賽事/課程動態(tài),可至翰林教育官網(wǎng)獲取更多信息。
往期文章閱讀推薦:
深耕九載!30+國際競賽/課程講義,碩博100%團(tuán)隊(duì)操刀,助力爬藤沖G5!

最新發(fā)布
? 2026. All Rights Reserved. 滬ICP備2023009024號-1









