- 翰林提供學術活動、國際課程、科研項目一站式留學背景提升服務!
- 021-63526630
Edexcel IGCSE Chemistry 復習筆記 2.1.2 Group 1: Reactivity & Electronic Configurations
Edexcel IGCSE Chemistry 復習筆記 2.1.2 Group 1: Reactivity & Electronic Configurations
Group 1: Reactivity & Electronic Configurations
- The reactivity of the group 1 metals?increases?as you go down the group
- When a group 1 element reacts its atoms only need to lose electron, as there is only 1 electron in the outer shell
- When this happens, 1+ ions are formed
- The next shell down automatically becomes the outermost shell and since it is?already full,?a group 1 ion obtains?noble gas configuration
- As you go down group 1, the number of shells of electrons increases by 1
- This means that the outermost electron gets?further?away from the nucleus, so there are?weaker?forces of attraction?between the outermost electron and the nucleus
- Less energy?is required to overcome the force of attraction as it gets weaker, so the outer electron is lost?more easily
- So, the alkali metals get more reactive as you descend the group
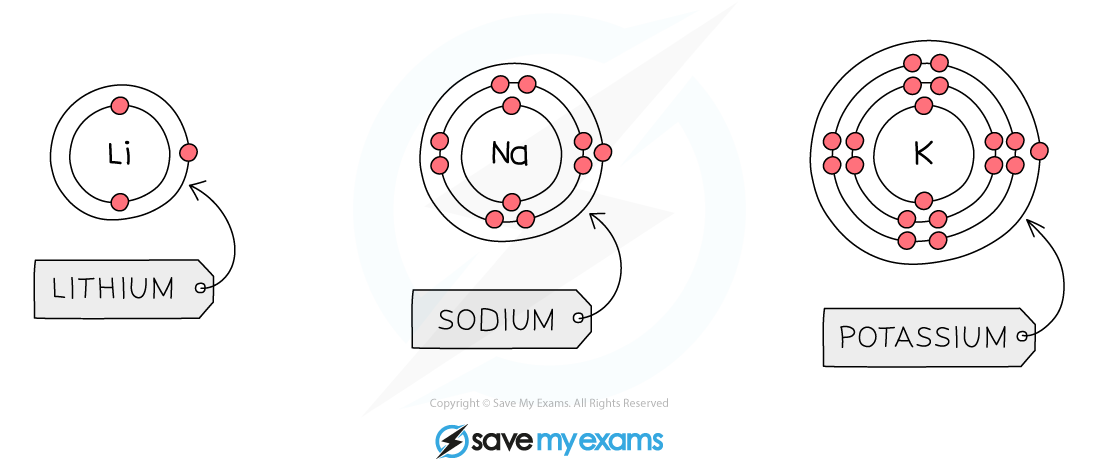
These electron shell diagrams of the first 3 alkali metals show that the group 1 metals have 1 electron in their outer shell
Exam Tip
In your exams, you could be asked to explain the trend in reactivity of the alkali metals - make sure you answer this question using their electronic configuration to support your answer.
轉載自savemyexam
以上就是關于【Edexcel IGCSE Chemistry 復習筆記 2.1.2 Group 1: Reactivity & Electronic Configurations】的解答,如需了解學校/賽事/課程動態,可至翰林教育官網獲取更多信息。
往期文章閱讀推薦:
深耕九載!30+國際競賽/課程講義,碩博100%團隊操刀,助力爬藤沖G5!

最新發布
? 2026. All Rights Reserved. 滬ICP備2023009024號-1









