- 翰林提供學術活動、國際課程、科研項目一站式留學背景提升服務!
- 021-63526630
CIE IGCSE Biology: 復習筆記:1.1.4 Diffusion
CIE IGCSE Biology: 復習筆記:1.1.4 Diffusion
Diffusion
- This is the process by which different gases or different liquids mix and is due to the random motion of their particles
- Diffusing particles move from an area of?high concentration?to an area of?low concentration
- Eventually the concentration of particles is?even?as they spread out to occupy all of the available space
- Diffusion happens on its own and no energy input is required although it occurs faster at higher temperatures
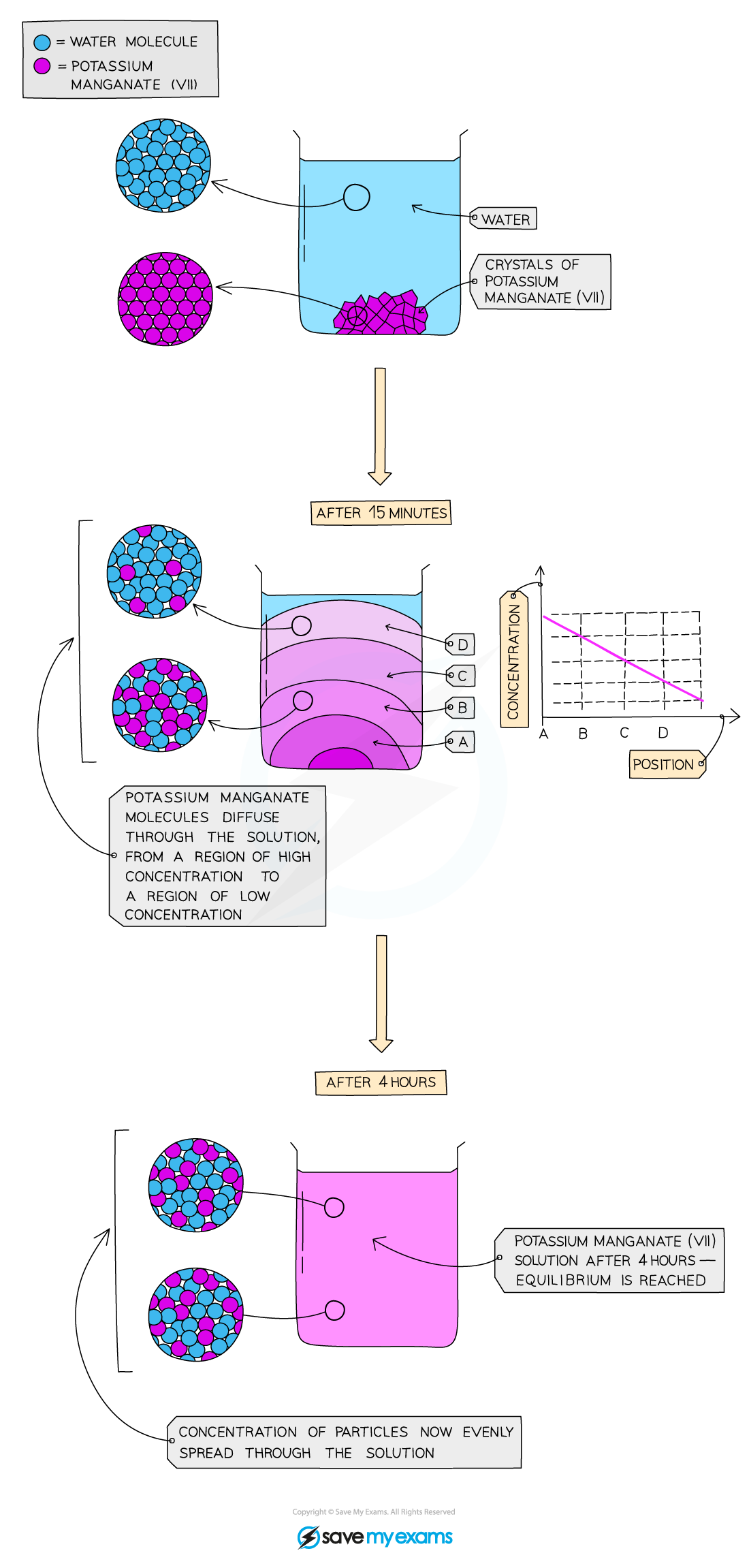
Diffusion of potassium manganate(VII), KMnO4?,?in water. After a few hours the concentration of KMnO4?is the same throughout the solution
Diffusion & Molecular Mass
EXTENDED
- Diffusion occurs much?faster?in gases than in liquids as gaseous particles move much quicker than liquid particles
- At the same temperature, different gases do not diffuse at the same rate.
- This is due to the difference in their?relative molecular masses
- Lighter gas particles can travel faster and hence further, therefore the lower its relative mass the faster a gas will diffuse
- This can be demonstrated in the reaction between ammonia, NH3, and hydrogen chloride gas, HCl, inside a long glass tube
- Where the two gases meet a white smoke of ammonium chloride, NH4Cl, is formed
- This does not occur in the middle of the tube as you might expect, but much closer to the end with the hydrogen chloride (Mr?= 36.5) and the ammonia (Mr?= 17) molecules are smaller and lighter
 NH3?molecules have less mass than the HCl molecule, so diffuse faster, hence the product (a white smoke of NH4Cl) forms closer to the end where the HCl is
NH3?molecules have less mass than the HCl molecule, so diffuse faster, hence the product (a white smoke of NH4Cl) forms closer to the end where the HCl is
轉載自savemyexams
以上就是關于【CIE IGCSE Biology: 復習筆記:1.1.4 Diffusion】的解答,如需了解學校/賽事/課程動態,可至翰林教育官網獲取更多信息。
往期文章閱讀推薦:
深耕九載!30+國際競賽/課程講義,碩博100%團隊操刀,助力爬藤沖G5!

最新發布
? 2026. All Rights Reserved. 滬ICP備2023009024號-1









