- 翰林提供學(xué)術(shù)活動、國際課程、科研項(xiàng)目一站式留學(xué)背景提升服務(wù)!
- 021-63526630
CIE A Level Chemistry復(fù)習(xí)筆記6.2.8 Coloured Complexes
Coloured Compounds & Electron Promotion
- Most transition element complexes are?coloured
- A transition element complex solution which is coloured,?absorbs?part of the electromagnetic spectrum in the visible light region
- The observed colour is the?complementary colour?which is made up of light with frequencies that are?not?absorbed
- For example, copper(II) ions absorb light from the?red?end of the spectrum
- The complementary colour observed is therefore?pale blue?(cyan)

The visible light region of the electromagnetic spectrum
Electron promotion
- In an?isolated?transition?element?ion?(which is not bonded to any ligands), all of the 3d orbitals are?degenerate
- However, when ligands are attached to the central metal ion through dative covalent bonds, these orbitals are split into two sets of?non-degenerate orbitals
- The difference in energy between these two sets of orbitals is?ΔE
- When light shines on a solution containing a transition element complex, an electron will absorb this exact amount of energy (ΔE)
- The amount of energy absorbed can be worked out by the equation:
ΔE?=?h?x?v
h?= Planck's constant (6.626 x 10-34?m2?kg s-1)
v?= frequency (Hertz, Hz or s-1)
- The electron uses the energy from the light to jump into a higher, non-degenerate energy level
- This is also called?electron promotion
- The other frequencies of light which are?not absorbed?combine to make the?complementary colour
- The diagram below shows an example of electron promotion in an octahedral complex of a nickel(II) Ni2+?ion
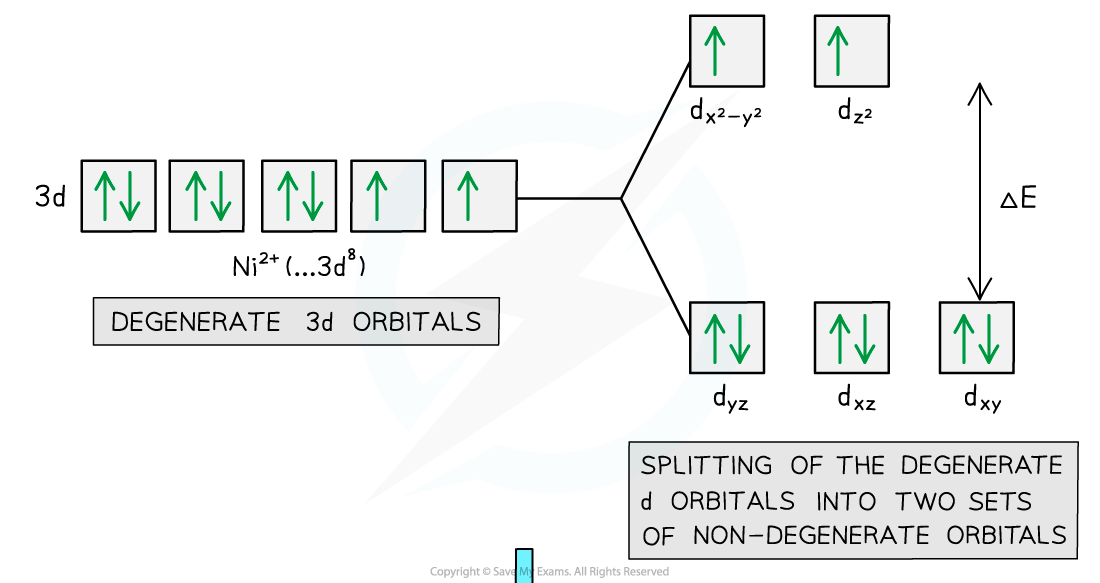
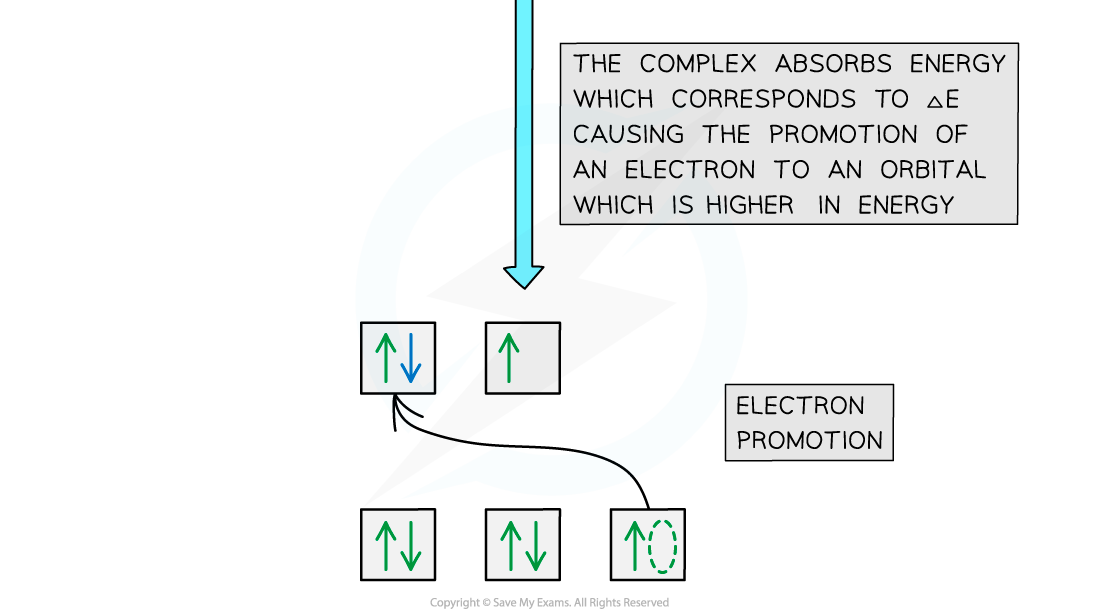
Electron promotion in a Ni(II) complex when light shines on the solution
Effects of Ligands on Complementary Colour
- Transition element complexes absorb the frequency of light which corresponds to the exact energy difference (ΔE) between their?non-degenerate?d orbitals
- The frequencies of light which are not absorbed combine to make the?complementary colour?of the complex
- It is the complementary colour which is seen
- However, the exact energy difference (ΔE) is affected by the different ligands which surround the transition element ion
- Different ligands will split the d orbital by a different amount of energy
- This depends on the?repulsion?that the d orbital experiences from these ligands
- Therefore, the?size?of?ΔE?and thus the?frequency?of light absorbed by the electrons will be slightly different
- As a result, a different colour of light is absorbed by the complex solution and a different?complementary colour?is observed
- This means that complexes with?similar?transition elements?ions,?but?different?ligands,?can have different colours
- For example, the [Cu(H2O)6]2+?complex has a?light blue?colour
- Whereas the [Cu(NH3)4?(H2O)2]2+?has a?dark blue?colour
- Despite the copper ion having an oxidation state of +2 in both complexes
- This is evidence that the ligands surrounding the?complex ion?affect the colour of the complex
Ligand Exchange in Copper(II) & Cobalt(II) Complexes
- Different ligands may affect the complementary colour of a transition ion complex solution
- This is shown by ligand exchange reactions in copper(II) and cobalt(II) complexes, as this causes a change in colour of the complexes
Copper(II) & cobalt(II) ions
- The ligand exchange of [Cu(H2O)6]2+?and [Co(H2O)6]2+?by NH3?ligands causes a change in the colour of the solutions
- [Cu(H2O)6]2+?is?light blue?in colour whereas [Cu(NH3)4(H2O)2)]2+?is?deep blue?in colour
- [Co(H2O)6]2+?is a?pink?solution whereas [Co(NH3)6]2+?is a?brown?solution
- The colour change results from the ammonia ligands, which cause the d orbitals to split by a different amount of energy (ΔE)
- Therefore, the size of?ΔE?and the frequency of light absorbed by the electrons will be slightly different
- As a result, a different colour of light is absorbed and thus a different?complementary colour?is observed

Ligand exchange of the water ligands by ammonia ligands causes a change in colour of the copper(II) complex solution
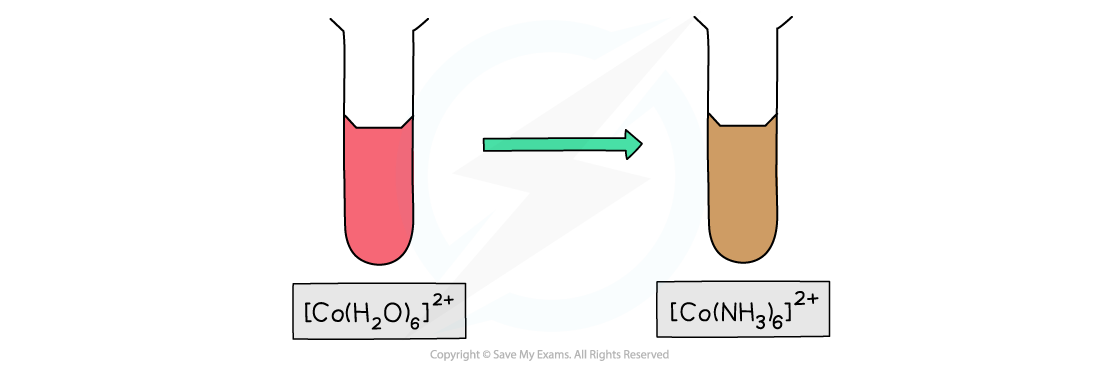
Ligand exchange of the water ligands by ammonia ligand causes a change in colour of the cobalt(II) complex solution
- Similarly, full ligand exchange by chloride ions in copper(II) and cobalt(II) complexes results in a change in complementary colour

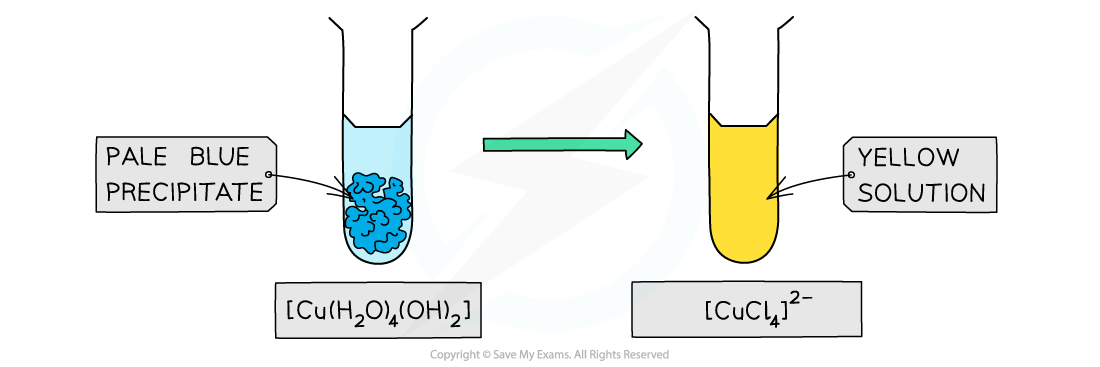
Ligand exchange by chloride ligands causes a change in colour of the copper(II) complex solution

Ligand exchange by chloride ligands causes a change in colour of the cobalt(II) complex solution
- As before, this suggests that different ligands will split the d orbitals differently
轉(zhuǎn)載自savemyexams
以上就是關(guān)于【CIE A Level Chemistry復(fù)習(xí)筆記6.2.8 Coloured Complexes】的解答,如需了解學(xué)校/賽事/課程動態(tài),可至翰林教育官網(wǎng)獲取更多信息。
往期文章閱讀推薦:
全網(wǎng)破防!ALevel CIE數(shù)學(xué)M1疑似錯題?經(jīng)濟(jì)P2難度飆升?5月6日大考考情分析必看!
A-Level CIE就大規(guī)模泄題發(fā)布最嚴(yán)處罰!哪些考生必須重考?你的成績怎么辦?

最新發(fā)布
? 2026. All Rights Reserved. 滬ICP備2023009024號-1









