- 翰林提供學(xué)術(shù)活動(dòng)、國(guó)際課程、科研項(xiàng)目一站式留學(xué)背景提升服務(wù)!
- 021-63526630
CIE A Level Chemistry復(fù)習(xí)筆記1.8.2 Rate of Reaction
Rate of Reaction: Concentration & Pressure
Concentration
- The more?concentrated?a solution is, the?greater?the number of?particles?in a given volume of solvent
- An increase in?concentration?causes in an increased?collision frequency?and therefore an increased?rate of reaction
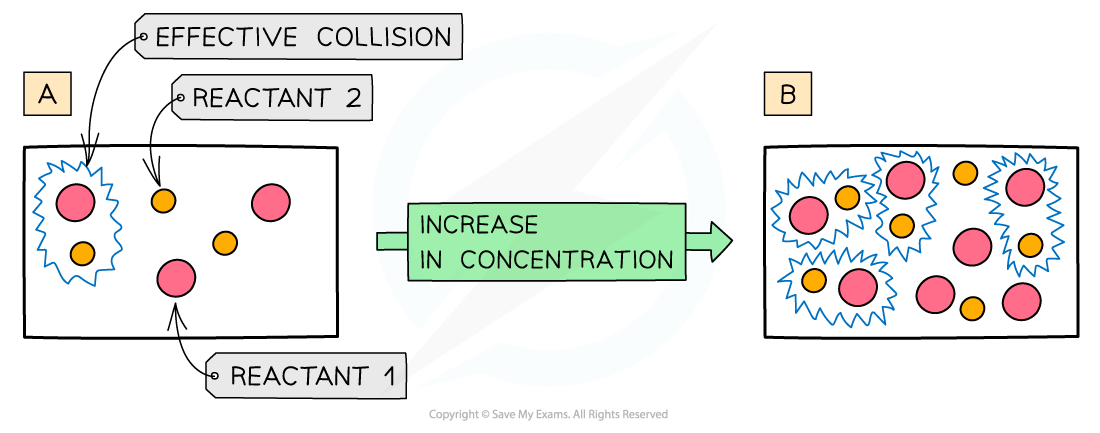
The diagram shows a higher concentration of particles in (b) which means that there are more particles present in the same volume than (a) so the chances and frequency of collisions between reacting particles is increased causing an increased rate of reaction
Pressure
- An increase in pressure in reactions that involve gases has the same effect as an increased concentration of solutions
- When the?pressure?is increased, the molecules have less space in which they can move
- This means that the number of?effective collisions?increases due to an increased?collision frequency
- An increase in pressure therefore increases the?rate of reaction
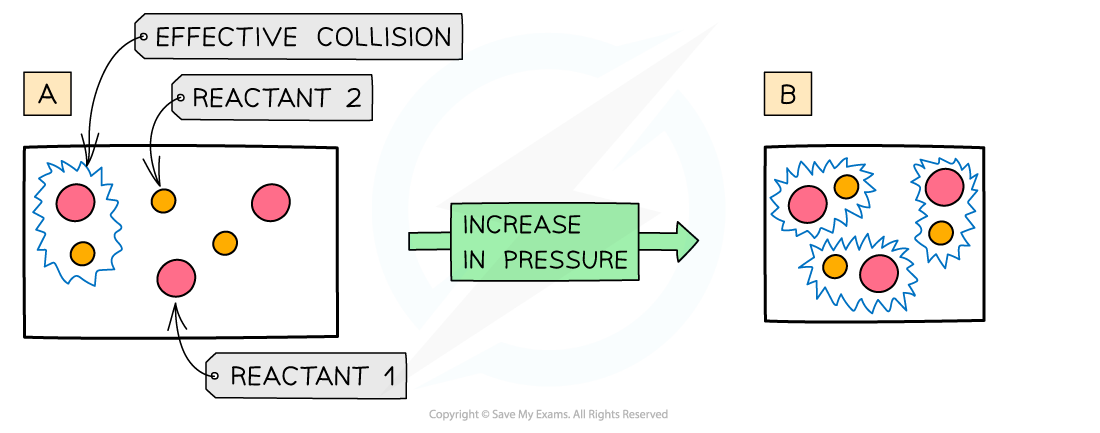
The diagram shows a higher pressure in (b) which means that the same number of particles occupy a smaller volume, resulting in an increased collision frequency and therefore increased rate of reaction
Rate of Reaction: Experimental Calculations
- During a reaction, the?reactants?are used up and changed into the?products
- This means that as the reaction proceeds, the concentration of the reactants is?decreasing?and the concentration of the products is?increasing
- Because of this, the?rate of the reaction?is not the same throughout the reaction but?changes
- The rate of reaction during the reaction can be calculated from a?concentration-time graph.
- The?isomerisation of cyclopropane?to propene will be taken as an example:

Isomerisation of cyclopropane
- The concentrations of reactant (cyclopropane) and product (propene) over time can be experimentally obtained
Concentrations of cyclopropane & propene table

- When taking the measurements, the?temperature?should be kept the same at all times as a?change?in temperature will change the?rate of reaction
- A?concentration-time graph?for the?concentration of propene as well as cyclopropane?can be obtained from the above results
- As an example, the concentration-time graph for propene is shown below:
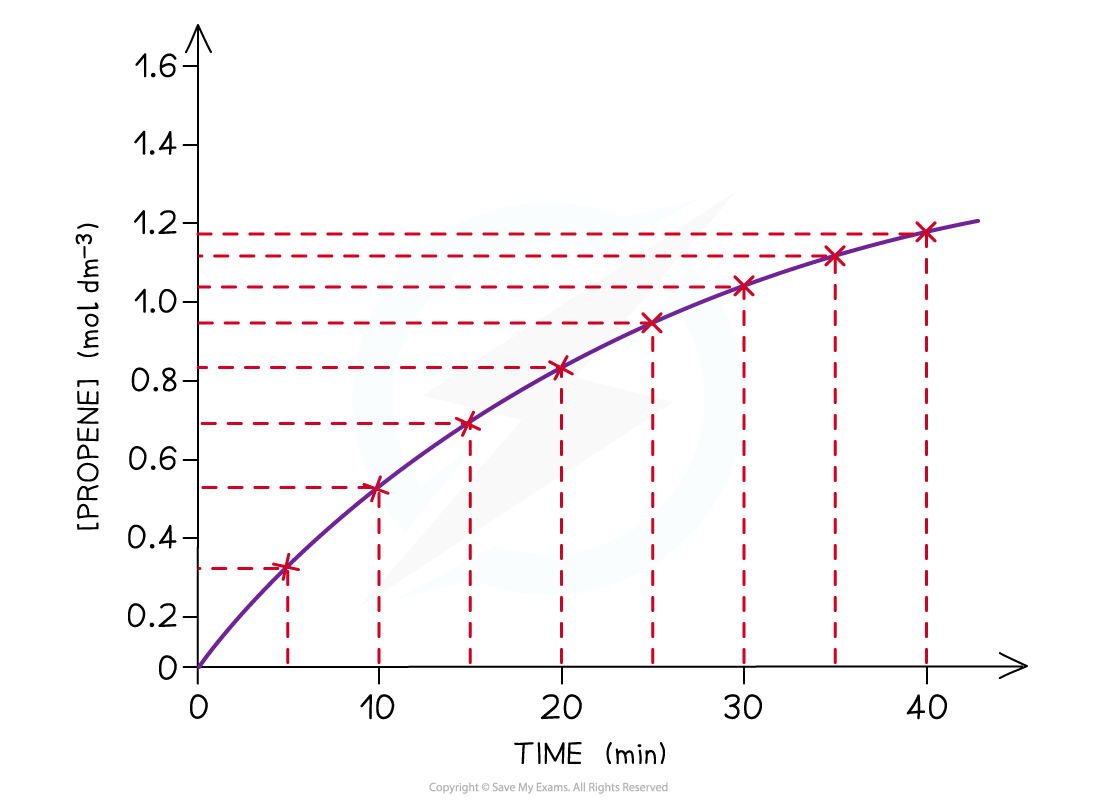 The graph shows that the concentration of propene increases with time
The graph shows that the concentration of propene increases with time
Calculating the rate at the start of a reaction
- At the start of the reaction, the concentration-time curve looks almost linear
- The rate at this point can therefore be found by treating the curve as a linear line and by using:

- Using the graph, the average rate of the reaction over the first 5 minutes for propene is:

Line a shows the average rate over the first five minutes whereas line b shows the actual initial rate found by drawing a tangent at the start of the curve. The calculated rates are very similar for both methods
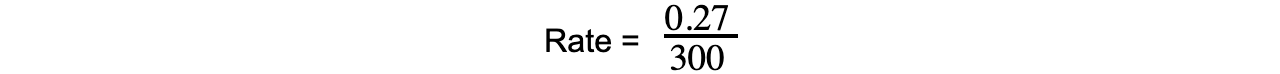
= 0.0009 mol dm-3?s-1
Calculating the rate as the reaction proceeds
- The curve becomes shallower with time which means that the rate decreases with time
- The rate of reaction can be calculated by taking short time intervals
- Eg. you can calculate the rate of reaction from 15 to 20 mins during which the concentration of propene increases from 0.68 to 0.83 mol dm-3:
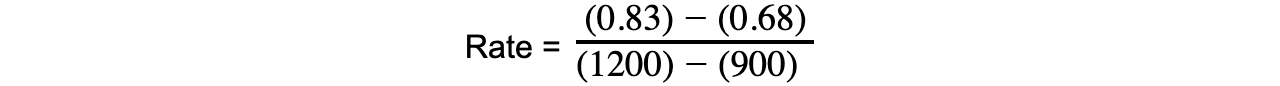
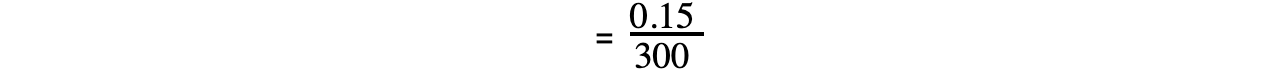
= 0.0005 mol dm-3 s-1
- The smaller the time intervals, the more accurate the reaction rate value is
- Even more accurate is to find the rate of reaction at?different concentrations?of reactant or product at?particular time?points
- This can be done by drawing?tangents?at several points on the graph
- As an example, the rates of reaction at different concentrations of cyclopropane are calculated by drawing the appropriate tangents:
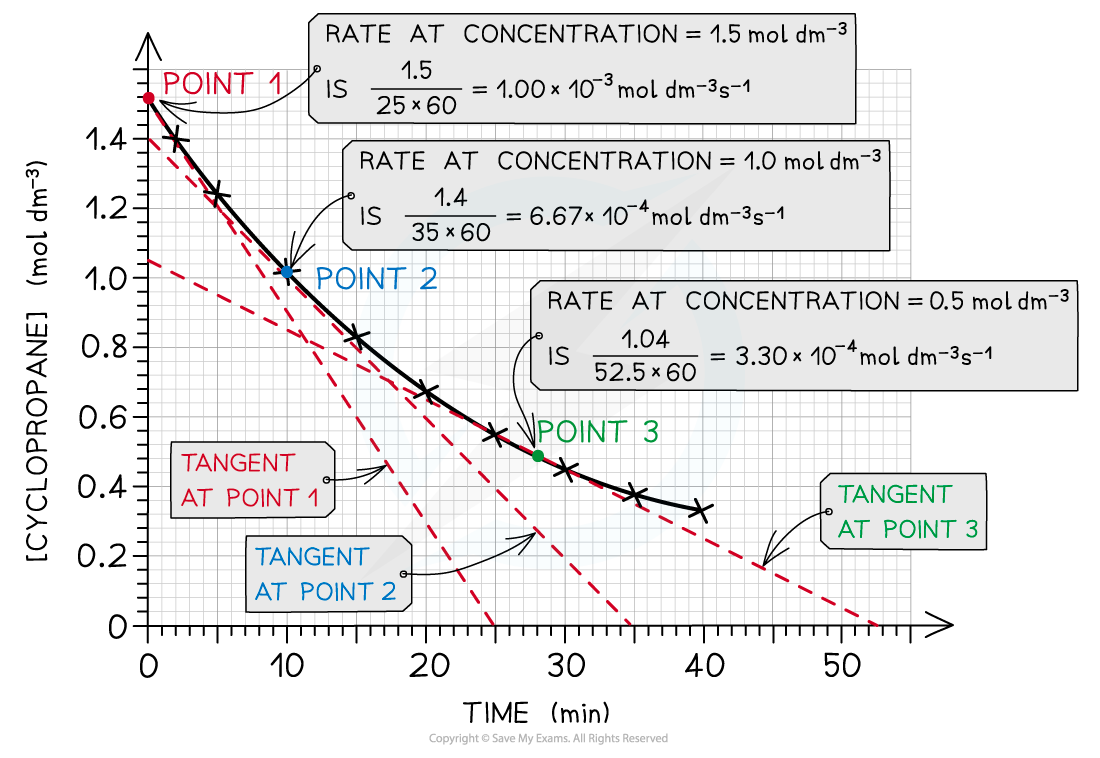
The rate of reaction at 3 different concentrations of cyclopropane is calculated by drawing tangents at those points in the graph
Rate-concentration graph
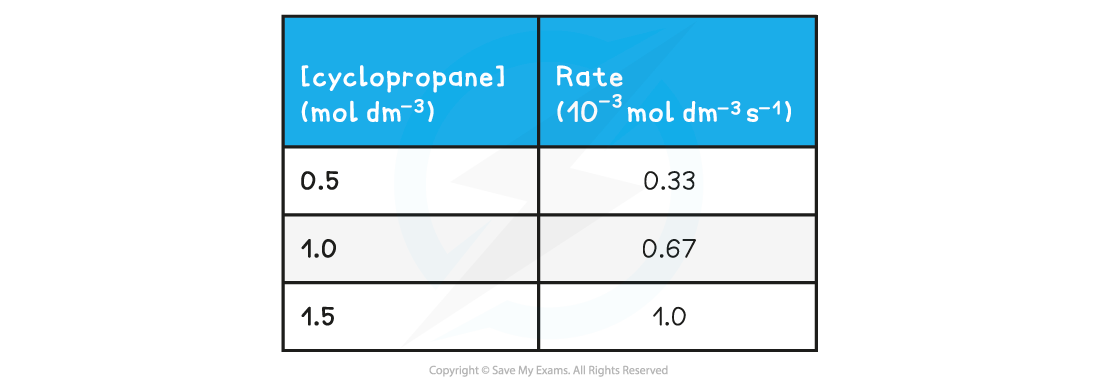
- The calculated rates can then be summarised in a table to show how the rate of reaction changes with changing concentration of the reactants or products
Change in rate with decreasing concentration of cyclopropane table
- This data can then be used to plot a rate-concentration graph
- The graph shows that the rate is directly proportional to the concentration of cyclopropane
- If you double the concentration of cyclopropane the rate of reaction will double too
 The graph shows a directly proportional correlation between the concentration of cyclopropane and the rate of reaction
The graph shows a directly proportional correlation between the concentration of cyclopropane and the rate of reaction
Exam Tip
To calculate the rate of reaction you can either use the increase in concentration of products (like in the example above) or the decrease in concentration of reactants.
轉(zhuǎn)載自savemyexams
以上就是關(guān)于【CIE A Level Chemistry復(fù)習(xí)筆記1.8.2 Rate of Reaction】的解答,如需了解學(xué)校/賽事/課程動(dòng)態(tài),可至翰林教育官網(wǎng)獲取更多信息。
往期文章閱讀推薦:
全網(wǎng)破防!ALevel CIE數(shù)學(xué)M1疑似錯(cuò)題?經(jīng)濟(jì)P2難度飆升?5月6日大考考情分析必看!
A-Level CIE就大規(guī)模泄題發(fā)布最嚴(yán)處罰!哪些考生必須重考?你的成績(jī)?cè)趺崔k?

最新發(fā)布
? 2026. All Rights Reserved. 滬ICP備2023009024號(hào)-1









