- 翰林提供學(xué)術(shù)活動(dòng)、國際課程、科研項(xiàng)目一站式留學(xué)背景提升服務(wù)!
- 021-63526630
CIE A Level Chemistry復(fù)習(xí)筆記1.6.3 Oxidising & Reducing Agents
Oxidising & Reducing Agents
Oxidising agent
- An oxidising agent?is a substance that?oxidises?another atom or ion by causing it to lose electrons
- An oxidising agent itself gets?reduced – gains electrons
- Therefore, the?ox. no.?of the oxidising agent?decreases

Example of an oxidising agent in a chemical reaction
Reducing agent
- A reducing agent?is a substance that?reduces?another atom or ion by causing it to gain electrons
- A reducing agent itself gets?oxidised – loses/donates electrons
- Therefore, the?ox.?no.?of the reducing agent?increases

Example of a reducing agent in a chemical reaction
- For a reaction to be recognised as a redox reaction, there must be both an oxidising and reducing agent
- Some substances can act both as oxidising and reducing agents
- Their nature is dependent upon what they are reacting with and the reaction conditions
Worked Example: Oxidising & reducing agents
Answer
Oxidising agents are substances that oxidise other species, gain electrons and are themselves reduced.
Write down the oxidation numbers of each species in the reaction
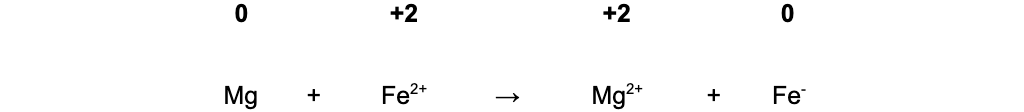
In equation B, Fe2+?oxidises Mg(0) to Mg2+(+2) and is itself reduced from Fe2+(+2) to Fe(0)
Roman numerals
- Roman numerals are used to show the?oxidation states of transition metals?which can have more than one oxidation number
- Iron can be both +2 and +3 so Roman numerals are used to distinguish between them
- Fe2+?in FeO can be written as?Iron(II) oxide
- Fe3+?in Fe2O3?can be written as?Iron(III) oxide
Worked example: Systematic names of compounds

Answer
Answer 1:?Iron(II) chloride: ox. no. of 2 Cl atoms is -2 and FeCl2?has overall no charge so ox. no. of Fe is +2
Answer 2:?Chloric(VII) acid: ox. no. of H is +1, 4 O atoms is -8 and HClO4?has overall no charge so ox. no. of Cl is +7
Answer 3:?Nitrogen(IV) oxide: ox. no. of 2 O atoms is -4 and NO2?has overall no charge so ox. no. of N is +4
Answer 4:?Magnesium nitrate: this is a salt of the common acid, so they are named without including the ox. no. of the non-metal
Answer 5:?Potassium sulfate: this is a salt of the common acid, so they are named without including the ox. no. of the non-metal
轉(zhuǎn)載自savemyexams
以上就是關(guān)于【CIE A Level Chemistry復(fù)習(xí)筆記1.6.3 Oxidising & Reducing Agents】的解答,如需了解學(xué)校/賽事/課程動(dòng)態(tài),可至翰林教育官網(wǎng)獲取更多信息。
往期文章閱讀推薦:
全網(wǎng)破防!ALevel CIE數(shù)學(xué)M1疑似錯(cuò)題?經(jīng)濟(jì)P2難度飆升?5月6日大考考情分析必看!
A-Level CIE就大規(guī)模泄題發(fā)布最嚴(yán)處罰!哪些考生必須重考?你的成績怎么辦?

最新發(fā)布
? 2026. All Rights Reserved. 滬ICP備2023009024號-1










