- 翰林提供學術活動、國際課程、科研項目一站式留學背景提升服務!
- 021-63526630
Edexcel IGCSE Chemistry 復習筆記 2.7.4 Bases
Edexcel IGCSE Chemistry 復習筆記 2.7.4 Bases
Bases
What makes a base act like a base?
Bases are substances which can neutralise an acid, forming a salt and water
The term base and alkali are not the same
So, all alkalis are bases, but not all bases are alkalisA base which is water-soluble is referred to as an alkali
Alkalis have pH values of above 7
In basic (alkaline) conditions red litmus paper turns blue
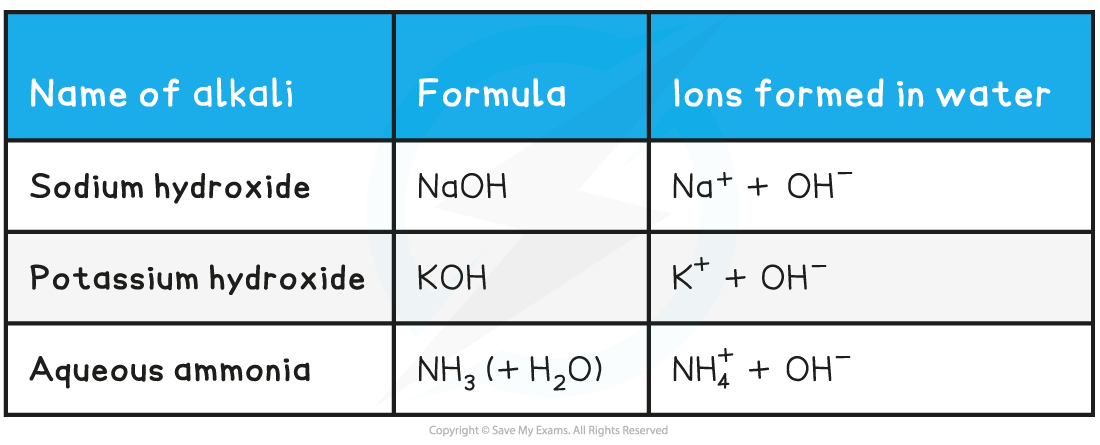
Bases are usually oxides, hydroxides or carbonates of metals
The presence of the OH- ions is what makes the aqueous solution an alkali
One unusual base is ammonia solution
When ammonia reacts with water it produces hydroxide ions
 ? Exam Tip
? Exam Tip轉載自savemyexam
以上就是關于【Edexcel IGCSE Chemistry 復習筆記 2.7.4 Bases】的解答,如需了解學校/賽事/課程動態,可至翰林教育官網獲取更多信息。
往期文章閱讀推薦:
深耕九載!30+國際競賽/課程講義,碩博100%團隊操刀,助力爬藤沖G5!

最新發布
? 2026. All Rights Reserved. 滬ICP備2023009024號-1









