- 翰林提供學(xué)術(shù)活動、國際課程、科研項(xiàng)目一站式留學(xué)背景提升服務(wù)!
- 021-63526630
Edexcel IGCSE Chemistry 復(fù)習(xí)筆記 2.6.2 Acids, Alkalis & Neutralisation
Edexcel IGCSE Chemistry 復(fù)習(xí)筆記 2.6.2 Acids, Alkalis & Neutralisation
Acids & Alkalis
- When?acids?are added to water, they form positively charged?hydrogen ions?(H+)
- The presence of H+?ions is what makes a solution acidic
- When?alkalis?are added to water, they form negative hydroxide ions (OH–)
- The presence of the OH–?ions is what makes the aqueous solution an alkali
- The pH scale is a numerical scale which is used to show how?acidic?or?alkaline?a solution is, in other words it is a measure of the amount of the ions present in solution
Neutralisation
- A neutralisation reaction occurs when an acid reacts with an alkali
- When these substances react together in a neutralisation reaction, the?H+?ions react with the?OH–?ions to produce water
- For example, when hydrochloric acid is neutralised a sodium chloride?and water are produced:
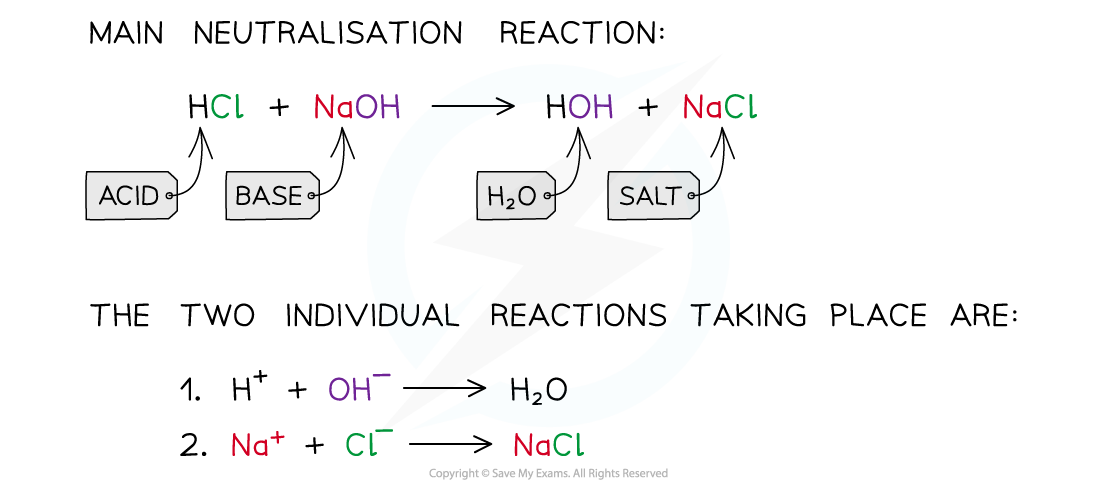
- The net ionic equation of?all acid-base neutralisations?and is what leads to a neutral solution, since water has a pH of 7:
H+??+??OH–?? H2O
- Neutralisation is very important in the treatment of soils to raise the pH as some crops cannot tolerate pH levels below 7
- This is achieved by adding bases to the soil such as limestone and quicklime
Exam Tip
Not all reactions of acids are neutralisations. For example, when a metal reacts with an acid, although a salt is produced there is no water formed so it does not fit the definition of neutralisation.
轉(zhuǎn)載自savemyexam
以上就是關(guān)于【Edexcel IGCSE Chemistry 復(fù)習(xí)筆記 2.6.2 Acids, Alkalis & Neutralisation】的解答,如需了解學(xué)校/賽事/課程動態(tài),可至翰林教育官網(wǎng)獲取更多信息。
往期文章閱讀推薦:
深耕九載!30+國際競賽/課程講義,碩博100%團(tuán)隊(duì)操刀,助力爬藤沖G5!

最新發(fā)布
? 2026. All Rights Reserved. 滬ICP備2023009024號-1









