- 翰林提供學(xué)術(shù)活動(dòng)、國際課程、科研項(xiàng)目一站式留學(xué)背景提升服務(wù)!
- 021-63526630
Edexcel IGCSE Chemistry 復(fù)習(xí)筆記 2.2.2 Group 7: Reactivity & Electronic Configurations
Edexcel IGCSE Chemistry 復(fù)習(xí)筆記 2.2.2 Group 7: Reactivity & Electronic Configurations
Group 7: Reactivity & Electronic Configurations
- When halogen atoms gain an electron during reactions, they form -1 ions called halide ions
- We can use electronic configuration to explain the trends in chemical reactivity down group 7
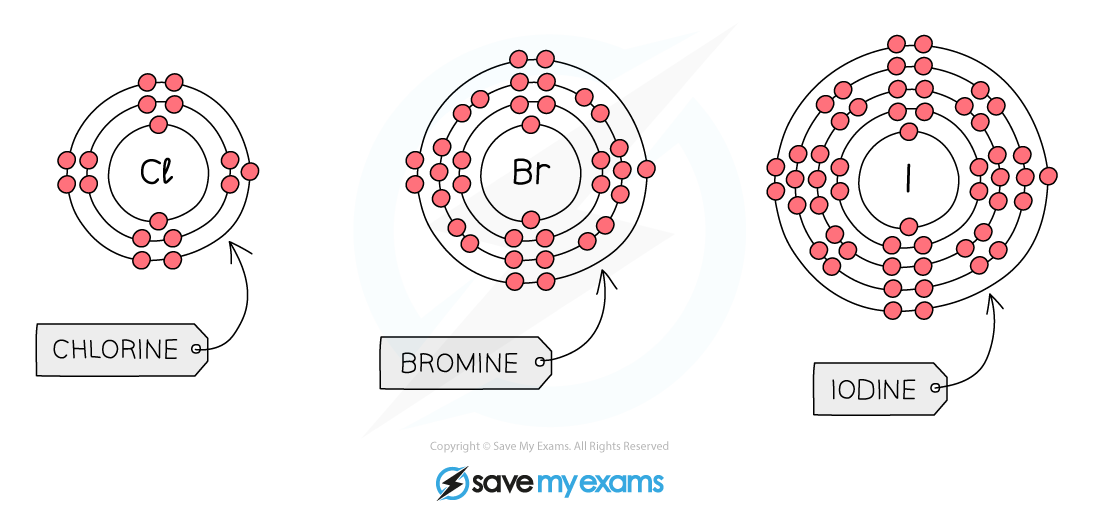
The atoms of the elements of group 7 all have 7 electrons in their outer shell
- Reactivity of group 7 non-metals?decreases?as you go down the group
- As you go down group 7, the number of shells of electrons?increases, the same as with all other groups
- However, halogen atoms form negative ions when they?gain an electron?to obtain a full outer shell
- This means that the increased distance from the outer shell to the nucleus as you go down a group makes the halogens become?less reactive
- Fluorine is the smallest halogen, which means its outermost shell is the?closest?to the positive nucleus of all the halogen
- Therefore, the ability to?attract an electron?is strongest in fluorine making it the most reactive
- As you move down the group, the forces of?attraction?between the nucleus and the outermost shell?decreases
- This makes it?harder?for the atoms to gain electrons as you descend the group
- Therefore, the halogens are less reactive the further down the group you go
Exam Tip
Exam questions on this topic occur often so make sure you know and can explain the reactivity trends of the group 7 elements in detail, using their electron configurations.
轉(zhuǎn)載自savemyexam
以上就是關(guān)于【Edexcel IGCSE Chemistry 復(fù)習(xí)筆記 2.2.2 Group 7: Reactivity & Electronic Configurations】的解答,如需了解學(xué)校/賽事/課程動(dòng)態(tài),可至翰林教育官網(wǎng)獲取更多信息。
往期文章閱讀推薦:
深耕九載!30+國際競(jìng)賽/課程講義,碩博100%團(tuán)隊(duì)操刀,助力爬藤?zèng)_G5!

最新發(fā)布
? 2026. All Rights Reserved. 滬ICP備2023009024號(hào)-1









