- 翰林提供學術活動、國際課程、科研項目一站式留學背景提升服務!
- 021-63526630
AQA A Level Physics復習筆記8.3.1 Nuclear Instability
Nuclear Stability Graph
- The most common elements in the universe all tend to have values of N and Z less than 20 (plus iron which has Z = 26, N = 30)
- Where:
- N = number of neutrons
- Z = number of protons / atomic number
- This is because lighter elements (with fewer protons) tend to be much?more stable?than heavier ones (with many protons)
- Nuclear stability becomes vastly clearer when viewed on a graph of N against Z
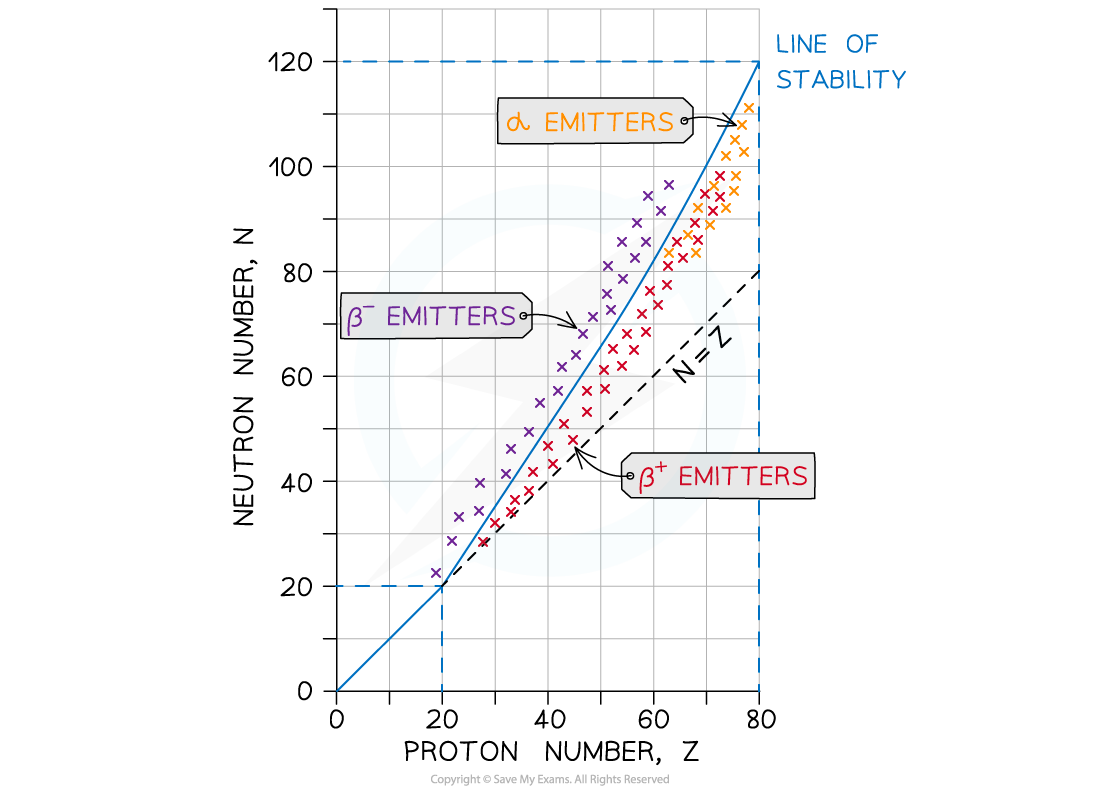
This nuclear stability curve shows the line of stable isotopes and which unstable isotopes will emit alpha or beta particles
- A nucleus will be unstable if it has:
- Too many neutrons
- Too many protons
- Too many nucleons ie. too heavy
- Too much energy
- An unstable atom wants to become neutral to become stable
- For light isotopes, Z < 20:
- All these nuclei tend to be very stable
- They follow the straight-line N = Z
- For heavy isotopes, Z > 20:
- The neutron-proton ratio increases
- Stable nuclei must have more neutrons than protons
- This imbalance in the neutron-proton ratio is very significant to the stability of nuclei
- At a short range (around 1–4 fm), nucleons are bound by the?strong nuclear force
- Below 1 fm, the strong nuclear force is?repulsive?in order to prevent the nucleus from collapsing
- At longer ranges, the electromagnetic force acts between protons, so?more protons cause more instability
- Therefore, as more protons are added to the nucleus, more neutrons are needed to add distance between protons to?reduce?the electrostatic repulsion
- Also, the extra neutrons increase the amount of binding force which helps to?bind the nucleons together
Alpha, Beta & Electron Capture
- The graph of N against Z is useful in determining which isotopes will decay via
- Alpha emission
- Beta-minus (β-) emission
- Beta-plus (β+) emission
- Electron capture
- Alpha-emitters:
- Occur beneath the line of stability when Z > 60 where there are?too many nucleons?in the nucleus
- These nuclei have more protons than neutrons, but they are too large to be stable
- This is because the strong nuclear force between the nucleons is unable to overcome the electrostatic force of repulsion between the protons
- Beta-minus (β-) emitters:
- Occur to the left of the stability line where the isotopes are?neutron-rich?compared to stable isotopes
- A neutron is converted to a proton and emits a β–?particle (and an anti-electron neutrino)
- Beta-plus (β+)?emitters:
- Occur to the right of the stability line where the isotopes are?proton-rich?compared to stable isotopes
- A proton is converted to a neutron and emits a β+?particle (and an electron neutrino)
- Electron capture:
- When a nucleus captures one of its own orbiting electrons
- As with β+?decay, a proton in the nucleus is converted into a neutron, releasing a gamma-ray (and an electron neutrino)
- Hence, this also occurs to the right of the stability line where the isotopes are?proton-rich?compared to stable isotopes
Exam Tip
To remember where the β-?and β+?emitters are on the graph:
- Beta-minus is a?negative particle where a?neutron turns into a proton. Unstable atoms always want to go towards a roughly equal number of protons and neutrons
- Therefore these emitters are on the?neutron-rich side of isotopes
- Beta-plus is a?positive particle where a?proton turns into a neutron
- Therefore these emitters are on the?proton-rich side of isotopes
The best way to remember the nuclear stability graph is to try to draw it from memory
轉載自savemyexams
以上就是關于【AQA A Level Physics復習筆記8.3.1 Nuclear Instability】的解答,如需了解學校/賽事/課程動態,可至翰林教育官網獲取更多信息。
往期文章閱讀推薦:
全網破防!ALevel CIE數學M1疑似錯題?經濟P2難度飆升?5月6日大考考情分析必看!
A-Level CIE就大規模泄題發布最嚴處罰!哪些考生必須重考?你的成績怎么辦?

最新發布
? 2026. All Rights Reserved. 滬ICP備2023009024號-1









