- 翰林提供學術活動、國際課程、科研項目一站式留學背景提升服務!
- 021-63526630
Edexcel IGCSE Chemistry 復習筆記 1.7.3 Simple Molecular Structures
Edexcel IGCSE Chemistry 復習筆記 1.7.3 Simple Molecular Structures
Simple Molecular Structures
- Simple molecular structures have covalent bonds joining the atoms together, but intermolecular forces that act between neighbouring molecules
- They have?low?melting and boiling points as there are only?weak intermolecular?forces acting?between?the molecules
- These forces are?very weak?when compared to the covalent bonds and so most small molecules are either gases or liquids at room temperature
- Often the liquids are volatile
- As the molecules increase in?size?the intermolecular forces also increase as there are more electrons available
- This causes the melting and boiling points to?increase
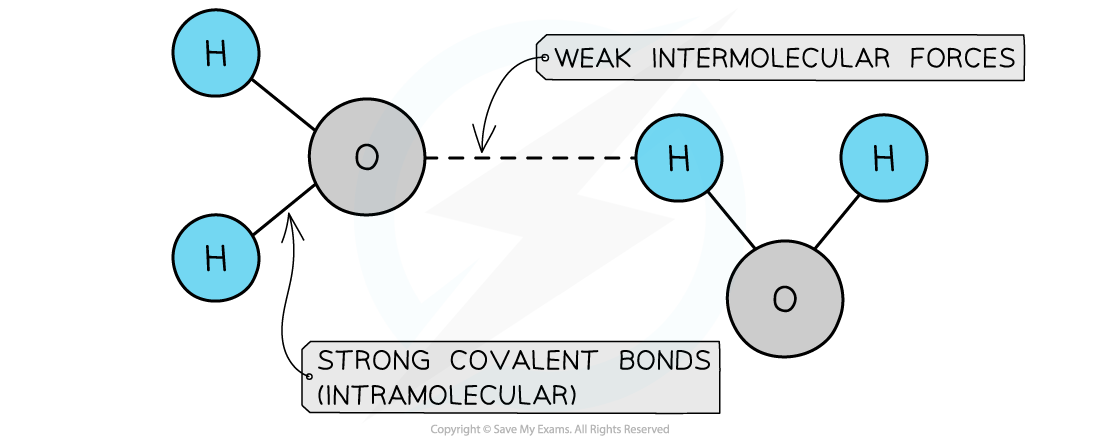
Covalent bonds are strong but intermolecular forces are weak
Exam Tip
The atoms within covalent molecules are held together by covalent bonds while the molecules in a covalent substance are attracted to each other by intermolecular forces.
Melting & Boiling Point Patterns
Melting and Boiling Point of Simple Compounds in Relation to Molecular Mass
- As the relative molecular mass of a substance increases, the melting and boiling point will increase as well
- An increase in the relative molecular mass of a substance means that there are more electrons in the structure, so there are more intermolecular forces of attraction that need to be overcome when a substance changes state
- So larger amounts of heat energy are needed to overcome these forces, causing the compound to have a higher melting and boiling point
- The family of organic molecules called alkanes show a clear increase in boiling point as the size of the molecule increases
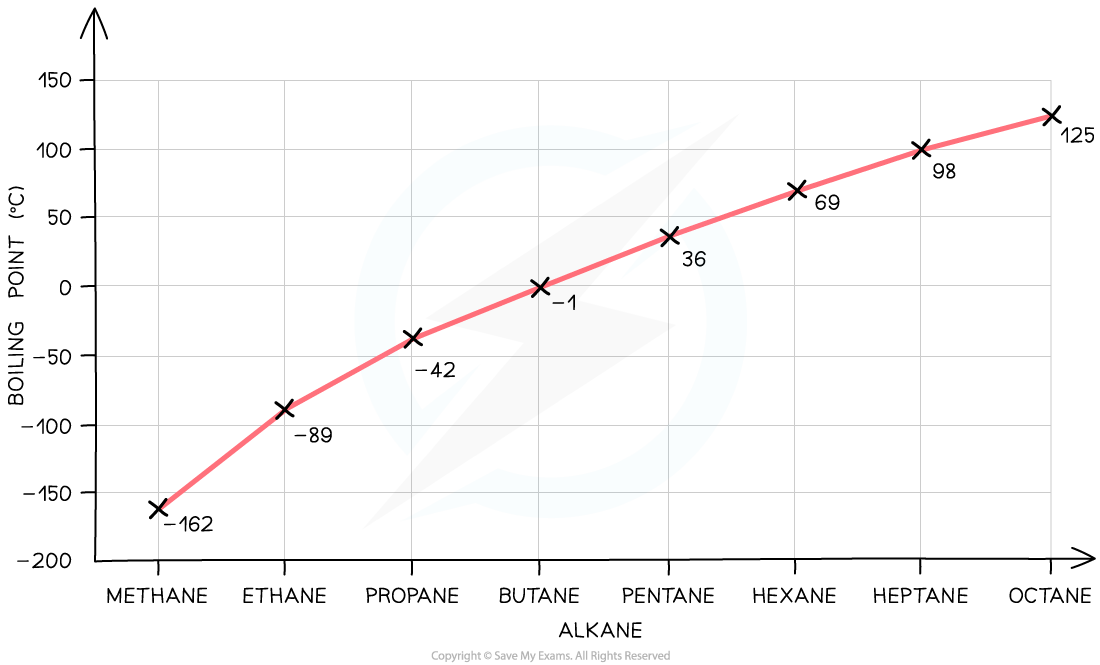
Graph?showing the increase in boiling point as the molecular size increases
Conductivity & Covalent Compounds
- They are?poor conductors?of electricity as there are no free ions or electrons to carry the charge
- Most covalent compounds do not conduct at all in the solid state and are thus?insulators
- Common insulators include the plastic coating around household electrical wiring, rubber and wood
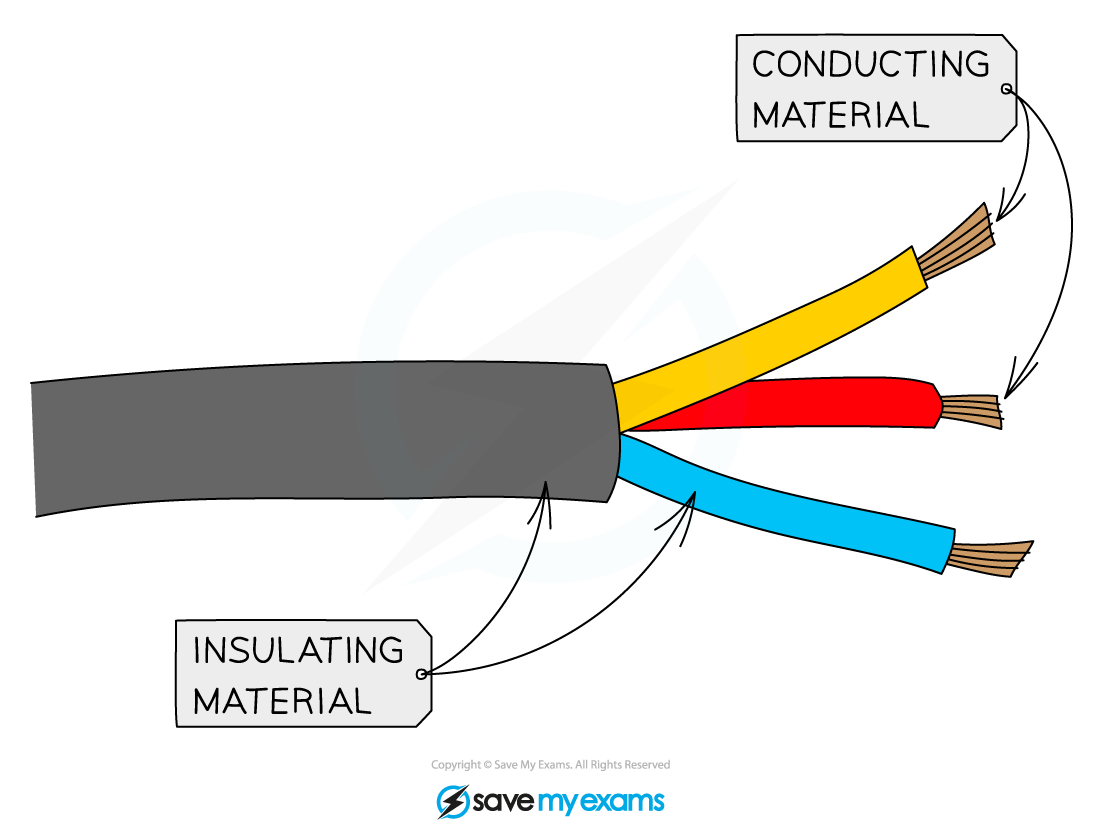
The plastic coating around electrical wires is made from covalent substances that do not allow a flow of charge
Exam Tip
When a covalent molecule melts or boils the covalent bonds do not break, only the intermolecular forces. If you think about it, when you boil a kettle full of water you are not generating large volumes of hydrogen and oxygen gas in your kitchen – this might give you an interesting unwanted chemical reaction ! Boom !
轉載自savemyexam
以上就是關于【Edexcel IGCSE Chemistry 復習筆記 1.7.3 Simple Molecular Structures】的解答,如需了解學校/賽事/課程動態,可至翰林教育官網獲取更多信息。
往期文章閱讀推薦:
深耕九載!30+國際競賽/課程講義,碩博100%團隊操刀,助力爬藤沖G5!

最新發布
? 2026. All Rights Reserved. 滬ICP備2023009024號-1









