- 翰林提供學術活動、國際課程、科研項目一站式留學背景提升服務!
- 021-63526630
Edexcel IGCSE Chemistry 復習筆記 1.7.1 Formation of Covalent Bonds
Edexcel IGCSE Chemistry 復習筆記 1.7.1 Formation of Covalent Bonds
Formation of Covalent Bonds
- Non-metal atoms can share electrons with other non-metal atoms to obtain a?full?outer?shell?of electrons
- When atoms share pairs of electrons, they form covalent bonds
- Covalent bonds between atoms are very?strong
- When two or more atoms are chemically bonded together, they form ‘molecules’
- Covalently bonded substances may consist of small molecules or giant molecules
- Weak?intermolecular forces?exist between individual molecules
- E.g. Each liquid water molecule consists of two hydrogen atoms covalently bonded to an oxygen atom, and in between two individual water molecules there are weak intermolecular forces
- Shared electrons are called?bonding electrons?and occur in?pairs
- Electrons on the outer shell which are not involved in the covalent bond(s) are called?non-bonding?electrons
- Simple covalent molecules do not conduct electricity as they do not contain free electrons

Diagram showing covalent bonding in a molecule of chlorine (Cl2)
Exam Tip
A key difference between covalent bonds and ionic bonds is that in covalent bonds the electrons are shared between the atoms, they are not transferred (donated or gained) and no ions are formed.
Electrostatic Attractions
- There is a strong electrostatic attraction between the shared pair of electrons and the nuclei of the atoms involved, since the electrons are negatively charged and the nuclei are positively charged
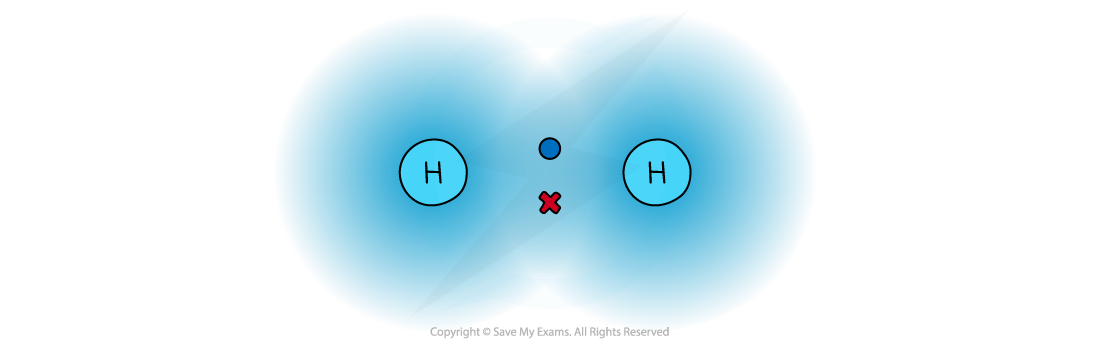
The attraction between the shared pair of electrons and the nuclei of the atoms involved in a covalent?bond
- In a normal covalent bond, each atom provide one of the electrons in the bond. A covalent bond is represented by a short straight line between the two atoms, H-H
- Covalent bonds should not be regarded as shared electron pairs in a fixed position; the electrons are in a state of constant motion and are best regarded as?charge clouds
- Sharing electrons in the covalent bond allows each of the 2 atoms to achieve an electron configuration similar to a noble gas
- This makes each atom more stable
轉載自savemyexam
以上就是關于【Edexcel IGCSE Chemistry 復習筆記 1.7.1 Formation of Covalent Bonds】的解答,如需了解學校/賽事/課程動態,可至翰林教育官網獲取更多信息。
往期文章閱讀推薦:
深耕九載!30+國際競賽/課程講義,碩博100%團隊操刀,助力爬藤沖G5!

最新發布
? 2026. All Rights Reserved. 滬ICP備2023009024號-1









