- 翰林提供學術活動、國際課程、科研項目一站式留學背景提升服務!
- 021-63526630
Edexcel IGCSE Chemistry 復習筆記 1.6 5 Ionic compounds: Bonds, Structure & Properties
Edexcel IGCSE Chemistry 復習筆記 1.6 5 Ionic compounds: Bonds, Structure & Properties
Ionic bonding
- The positive and negative charges are held together by the strong?electrostatic forces of?attraction?between?oppositely?charged ions
- This is what holds ionic compounds together
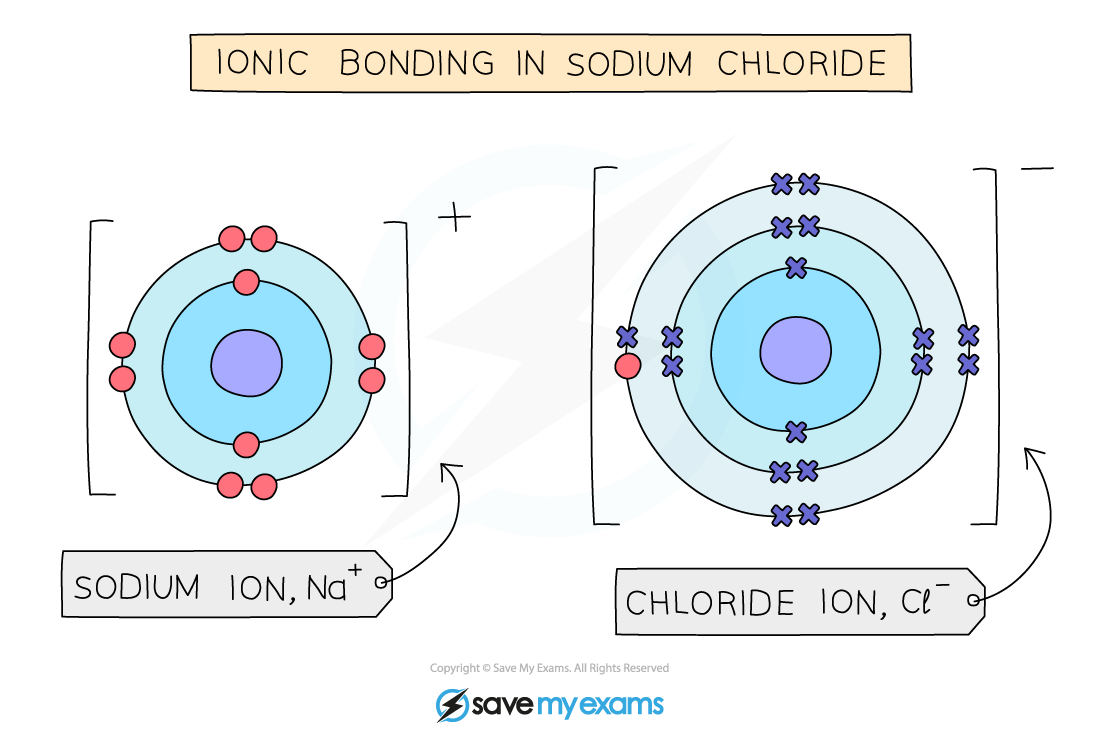
Electrostatic forces hold the ions together in sodium chloride
Giant Ionic Lattices
- Ionic compounds are made of charged particles called ions which form a giant lattice structure
- Ionic substances have?high?melting and boiling points due to the presence of?strong?electrostatic forces?acting between the oppositely charged ions
- These forces act in all directions and a lot of energy is required to overcome them

Strong electrostatic forces act in all directions in an ionic solid such as sodium chloride
- Ionic compounds are usually?solid?at room temperature and are?non-volatile
- They are usually?water soluble?as both ionic compounds and water are?polar??substances
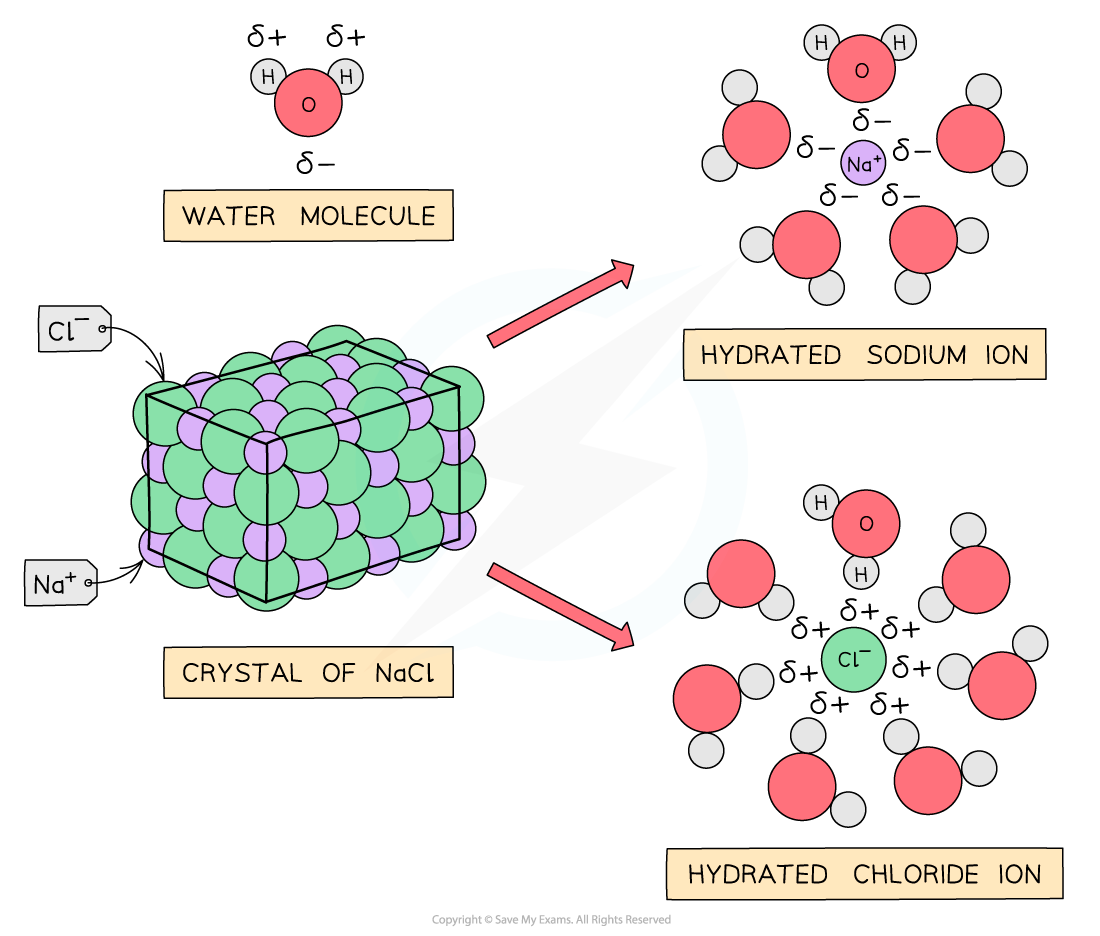
Ionic compounds are soluble in water because the ions are easily hydrated by polar water molecules
Exam Tip
Ions with higher charge have stronger electrostatic forces and will thus have higher melting and boiling points.
Conductivity & Ionic Compounds
- For electrical current to flow there must be present freely moving charged particles such as electrons or ions
- Ionic compounds can conduct electricity in the?molten?state or in?solution?as they have ions that can move and carry charge
- They cannot conduct electricity in the solid state as the ions are in fixed positions within the lattice and are unable to move
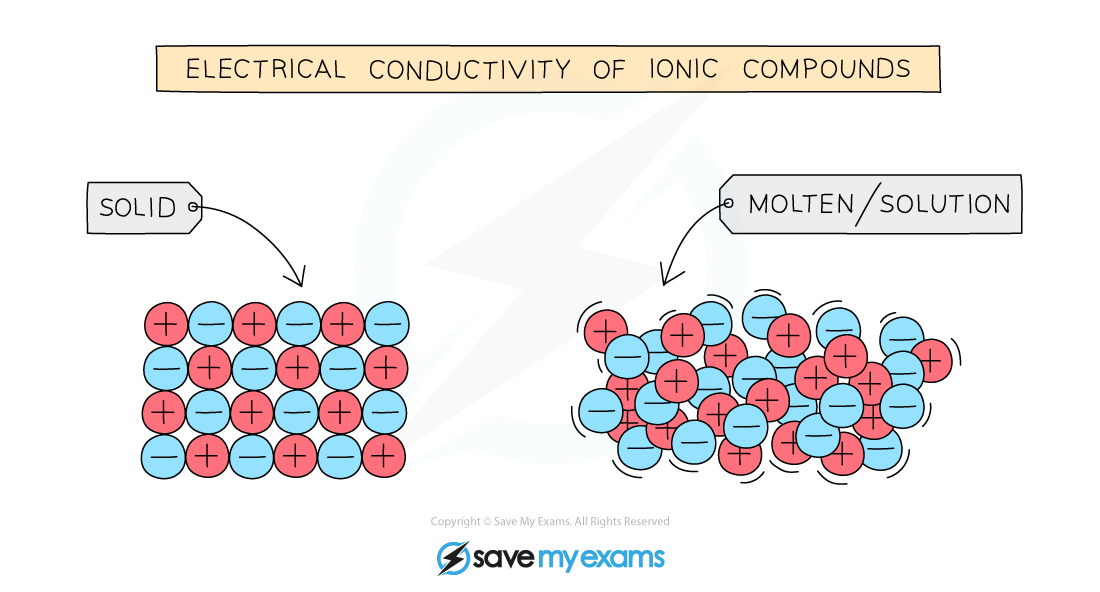
Molten or aqueous particles move and conduct electricity but cannot in the solid state
Exam Tip
Remember that in ionic lattice structures, positively charged and negatively charged ions are arranged in an alternating pattern.
轉載自savemyexam
以上就是關于【Edexcel IGCSE Chemistry 復習筆記 1.6 5 Ionic compounds: Bonds, Structure & Properties】的解答,如需了解學校/賽事/課程動態,可至翰林教育官網獲取更多信息。
往期文章閱讀推薦:
深耕九載!30+國際競賽/課程講義,碩博100%團隊操刀,助力爬藤沖G5!

最新發布
? 2026. All Rights Reserved. 滬ICP備2023009024號-1









